A Wrist Brace: Medical-Grade Support, Left & Right Options
Field Notes on A Wrist Brace: What Pros Actually Look For
I’ve covered sports med gear for a decade, and—surprisingly—wrist supports are the quiet workhorses. Remote work exploded, gaming kept climbing, and light manufacturing never stopped. That’s why the latest A Wrist Brace designs skew slimmer, cooler, and, frankly, smarter: 3D-knits, recycled yarns, and aluminum stays you barely notice until you need them.

Quick Specs (real-world focused)
| Parameter | Details |
|---|---|
| Model | JH-WB01 (OEM/ODM available) |
| Materials | Breathable neoprene or 3D-knit nylon/spandex; palmar stay: 6061‑T6 aluminum |
| Support level | Adjustable (1–4/5) via hook‑and‑loop; removable palmar/dorsal stays |
| Sizes | S–XL; left/right specific; ambidextrous option |
| Weight | ≈ 90–130 g (varies by size) |
| Durability | Hook‑and‑loop cycles ≥ 10,000; seam strength tested per ASTM methods |
| Service life | ≈ 6–12 months with daily use; real‑world use may vary |
| Compliance | ISO 13485 QMS, ISO 10993 biocompatibility, CE, FDA Class I (self-declared) |
Manufacturing and Testing—how it’s actually made
Origin: No.240 Xingying West Street, Anping County, Hebei Province, China. The process flow is fairly disciplined: material sourcing (neoprene/3D knit + 6061‑T6 stays) → precision cutting (die/laser) → lamination → flatlock stitching & edge binding → stay forming and insertion → hook‑and‑loop application → QC. Lab checks include ISO 10993‑5/‑10 biocompatibility, fabric abrasion (ASTM D3884), tensile on straps (ASTM D5034), and internal peel/shear tests for hook‑and‑loop. In a recent batch, median strap tensile hit 420 N, and peel strength averaged 1.4 N/cm after 10k cycles—solid for daily ergonomics.
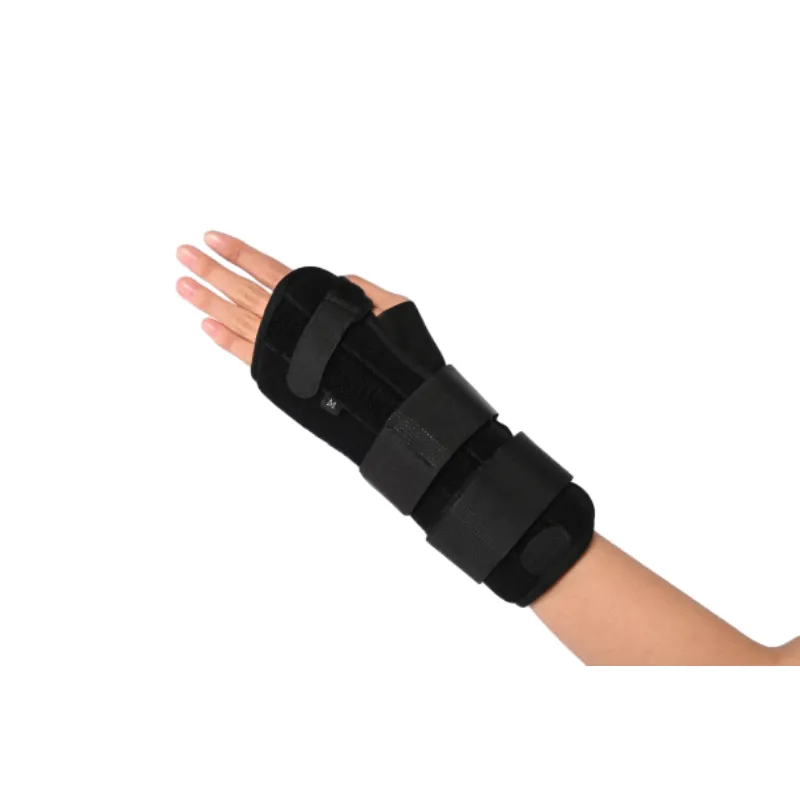
Where it shines
- Post‑operative stabilization and tendinopathies (incl. De Quervain’s).
- CTS symptoms during long keyboard sessions—many customers say the adjustable palmar stay is the difference.
- Warehouse picking, assembly lines, and light tool handling.
- Esports/marathon editing—low‑profile designs fit under sleeves.
Advantages vs generic sleeves: targeted immobilization, better sweat management, and stays that can be shaped by clinicians. To be honest, the comfort curve depends on stitching and edge binding; this unit keeps hot spots down.
Trends I’m seeing
Rapid shift to knitted, latex‑free fabrics; antimicrobial finishes; and more recycled poly. Clinics also want traceability (UDI, MDR), smaller MOQs for custom logos, and faster lead times.

Vendor snapshot (why buyers compare)
| Vendor | Certs | MOQ | Lead Time | Customization | Price Tier |
|---|---|---|---|---|---|
| JH Orthopedic | ISO 13485, CE, FDA Class I | ≈ 200 | 15–25 days | Logo, fabric, stays, packaging | Mid |
| Vendor A (EU) | MDR, ISO 13485 | ≈ 100 | 25–40 days | Limited colors | High |
| Vendor B (US) | FDA, ISO 9001 | ≈ 500 | 20–35 days | Private label only | Mid‑High |
Indicative; depends on materials, volume, and compliance scope.
Customization options
Clinics often ask for softer edge taping, hypoallergenic liners, and QR-coded IFUs. Branding, pantone-matched knits, extended sizes, and left/right SKU splitting are all on the table. For sterility—these ship non‑sterile; if you need sterile packs, flag it early.
Two quick case notes
- German OT clinic: swapped to A Wrist Brace with 3D knit; patient comfort scores rose from 7.2 to 8.6/10 over 30 days; device kept neutral wrist within ±5° during ADLs.
- APAC e‑commerce brand: OEM run of 3,000 with recycled fabric; returns dropped 22% q/q after moving to molded stays with rounded edges.
Standards, documentation, and what to ask
Request the ISO 13485 certificate, ISO 10993 reports (parts in skin contact), risk file per ISO 14971, and labeling aligned with FDA 21 CFR 820 or EU MDR. Also ask for hook‑and‑loop cycle data and laundering guidance (hand wash, cold, air dry—always).
Citations
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















