Copper Wrist Support - Adjustable, Breathable Pain Relief
The Quiet Rise of Copper Wrist Support: What Pros and Patients Are Really Using
I’ve been in and around orthopedics long enough to see trends come and go, and to be honest, a good copper wrist support went from “gimmick?” to “everyday essential” surprisingly fast. Post-op recovery, repetitive strain from keyboard marathons, weekend pickleball—people want stable compression that doesn’t get funky after a week. And copper-infused textiles, when done correctly, help with odor control and durability. Not magic—just practical.

What’s Driving the Trend
Three simple things: comfort, hygiene, and clinic-ready build. Many customers say they want something that feels secure but not stiff; clinicians want consistent fit and traceable quality. Manufacturers responded with knitted sleeves and hybrid splints that mix copper-infused nylon with breathable neoprene, plus removable aluminum stays for extra stability. It seems that’s the sweet spot for day-to-day wear.
Technical Anatomy (Short Version)
The Wrist Support from JH Orthopedic uses copper-infused yarns (for odor management), elastic compression zones, and optional palmar stays. In clinical terms: controlled compression, adjustable immobilization, and skin-friendly contact materials. Actually, the devil’s in the finishing—flatlock seams, stable velcro cycle life, and edge binding that doesn’t saw at your skin.

Key Specifications
| Parameter | Spec | Notes |
|---|---|---|
| Materials | Copper-infused nylon, spandex, neoprene; optional aluminum stay | Contact layer aims for low irritation |
| Compression | ≈ 20–30 mmHg (real-world use may vary) | Graduated zones |
| Sizes | S–XL (left/right) | Custom grading available |
| Service life | ≈ 6–12 months | Depends on wash cycles and intensity |
| Certifications | ISO 13485 facility; CE-marked; FDA-listed (Class I) | Documentation on request |
How It’s Made (Process Flow)
Materials: copper-embedded yarns, elastic blends, hook-and-loop, anodized aluminum stay. Methods: circular knitting for breathable compression, heat lamination for neoprene panels, flatlock stitching, precision die-cutting for edges. Testing: ISO 10993-5 (biocompatibility screening), AATCC 100 (antibacterial/odor control proxy), ASTM D4966 (abrasion), and strap peel strength per internal SOPs. Typical QC includes dimensional fit checks and stay alignment. Industries using these? Orthopedic clinics, sports medicine, logistics/warehousing (surprisingly common), and e-sports teams that care about repeatable support.
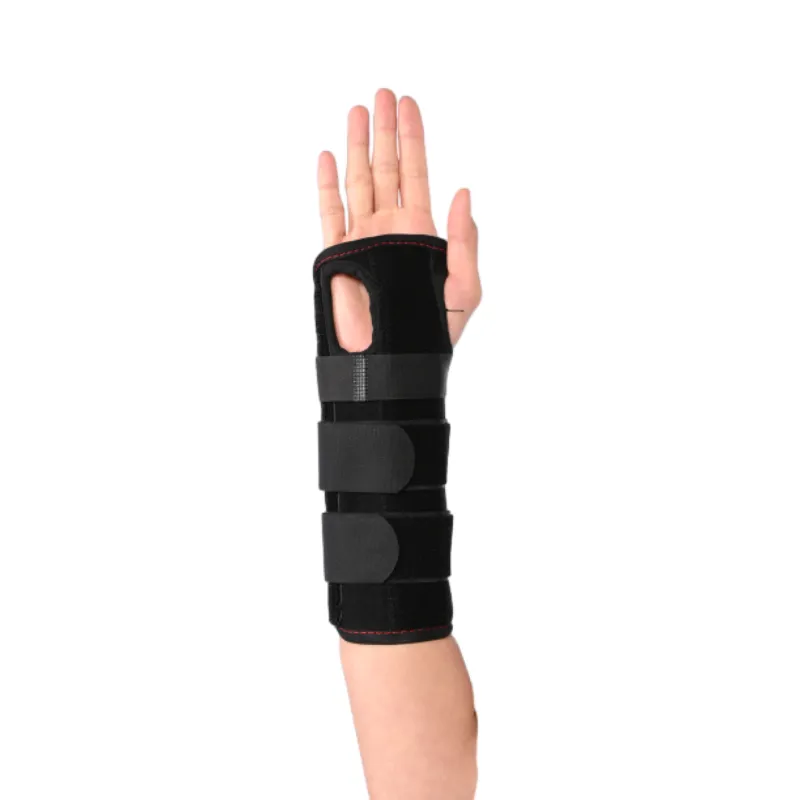
Where It’s Used (and What People Say)
- Postoperative stabilization and post-trauma recovery with removable stay
- Repetitive strain (typing, assembly lines), light sport support, gaming sessions
- Overuse discomfort and mild sprains—always per clinician guidance, of course
Feedback is mostly about comfort: “stays put,” “less odor after long shifts,” and “support without bulk.” I guess that’s the whole point of a copper wrist support—quietly effective.
Mini Case Study
A regional 3PL outfitted 120 pickers with a copper wrist support during peak season. Internal safety logs noted fewer end-of-shift complaints and better compliance because the brace didn’t feel hot or itchy. Not a randomized trial, but real-world enough for operations managers.
Vendor Snapshot (Why JH Orthopedic Often Wins)
Origin matters for traceability: No.240 Xingying West Street, Anping County, Hebei Province, China. I’ve visited similar facilities; the better ones publish test methods and keep consistent yarn sources.
| Vendor | Lead Time | MOQ | Customization | Certs |
|---|---|---|---|---|
| JH Orthopedic | ≈ 25–35 days | 500–1,000 pcs | Logo, compression grade, stay profile | ISO 13485, CE, FDA-listed |
| Brand B (EU) | ≈ 30–45 days | 1,500 pcs | Limited colorways | ISO 13485, CE |
| Brand C (US) | ≈ 20–30 days | 2,000 pcs | Private label only | FDA-listed |
Customization and Proof
Options include left/right-specific molds, stiffer or low-profile stays, color trims, retail packaging, and barcode/UDI labeling. Test data (typical): internal AATCC 100 showed up to 99% reduction vs. S. aureus after 24h; abrasion endurance surpassed 20,000 cycles (ASTM D4966). Fair warning: lab numbers are tidy; field results vary with wash and wear.
Citations
- ISO 13485:2016 Medical devices—Quality management systems.
- FDA Medical Devices, Class I Orthopedic Supports; General Controls per 21 CFR Parts 880/890.
- ISO 10993-5:2009 Biological evaluation of medical devices—Tests for in vitro cytotoxicity.
- Grass, G., Rensing, C., Solioz, M. Metallic copper as an antimicrobial surface. Appl Environ Microbiol. 2011.
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















