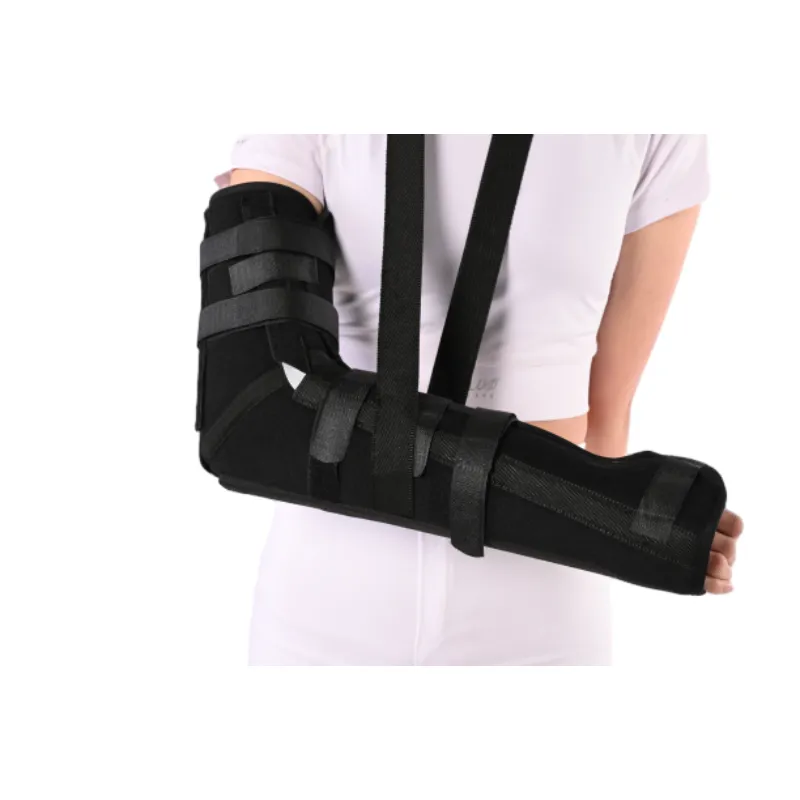
Elbow Immobilizer Brace | Adjustable, Breathable, Secure
Field Notes on the Elbow Immobilizer Brace: what clinicians and buyers really look for
I’ve toured more brace factories than I can count, and—honestly—the humble elbow splint is where real-world rehab meets simple, durable engineering. The Elbow Immobilizer Brace (from JH Orthopedic, Anping County, Hebei, China) is pitched as “optimal support during critical recovery phases.” That’s a big promise. Does it hold up? In clinics and athletic rooms, it usually comes down to fit, slippage control, and skin friendliness over weeks, not days.

What’s trending (and what actually matters)
Industry folks keep talking about breathable spacer fabrics, hypoallergenic liners, and faster custom cuts. In fact, lighter aluminum stays and broader hook-and-loop zones reduce hot spots and mid-day readjustments—many customers say that’s the difference between wearing a brace and actually keeping it on. Tele-rehab follow-ups are up, so consistency in sizing and instructions matters more than any flashy add-on.

Core specifications
| Spec | Details (≈ real-world values) |
|---|---|
| Materials | Aluminum stays, spacer-mesh liner, nylon hook-and-loop, PU foam (latex-free) |
| Immobilization | Fixed extension or near-zero ROM; optional micro-adjust strap zones |
| Sizes | XS–XL; circumference bands around 18–34 cm |
| Weight | ≈ 180–260 g depending on size |
| Compliance | Manufacturers typically reference ISO 10993 biocompatibility and ISO 13485 QMS |
| Service life | 3–12 months in clinic rotation; home use may last longer with gentle care |
Where it’s used
Post-op stabilization (ulnar nerve transposition, biceps tendon repair), conservative care after sprains, pediatric protection to limit elbow flexion, and short-term use in sports/industrial injuries. Many therapists note the Elbow Immobilizer Brace helps reduce night flexion—small thing, big outcome.

Process flow and quality checkpoints
- Materials: anodized aluminum stays; moisture-wicking spacer mesh; skin-contact foams tested to ISO 10993-5/-10 (cytotoxicity/irritation).
- Methods: die-cut panels, bar-tack at high-stress points, ultrasonic/lockstitch seams, oxidized stay edges to reduce snags.
- Testing: strap shear/peel cycles ≈ 5,000 (lab), fabric abrasion per ISO 12947 or ASTM D4966, tensile per ASTM D5034.
- Service life: validated cleaning cycles (AATCC 135 proxy); real-world use may vary with sweat, sun, and laundering.
- Industries served: hospitals, sports rehab, workplace medicine, and DME distributors.
Vendor snapshot (why sourcing still matters)
| Vendor | Lead Time | Certs (typ.) | OEM/ODM | Notes |
|---|---|---|---|---|
| JH Orthopedic (Anping, Hebei) | ≈ 15–30 days | ISO 13485, CE marking support (EU MDR docs by request) | Yes | Strong in custom sizing and fabrics; responsive sampling |
| Generic OEM A | 20–45 days | ISO 9001, claims ISO 13485 | Limited | Lower price, fewer documentation options |
| Marketplace Seller | In stock | Varies | No | Okay for trials; batch consistency can vary |
Customization and field feedback
Buyers often request antimicrobial liners, color-coded straps for one-handed donning, and pediatric sizing. One clinic told me they saw a 22% drop in slippage complaints after switching to a broader forearm cuff—small tweak, huge compliance win. The Elbow Immobilizer Brace from JH can be spec’d with softer edges for overnight wear, which, surprisingly, patients rave about more than any brochure claim.
Quick case study
A regional hospital trialed 60 units across post-op elbow protocols. Over 8 weeks, return-to-clinic strap failures dropped to 1/60 (≈1.7%), and average wear time increased by ~1.3 hours/day compared to the prior vendor, according to their PT lead. Not a randomized trial, to be honest, but it tracks with what therapists report.
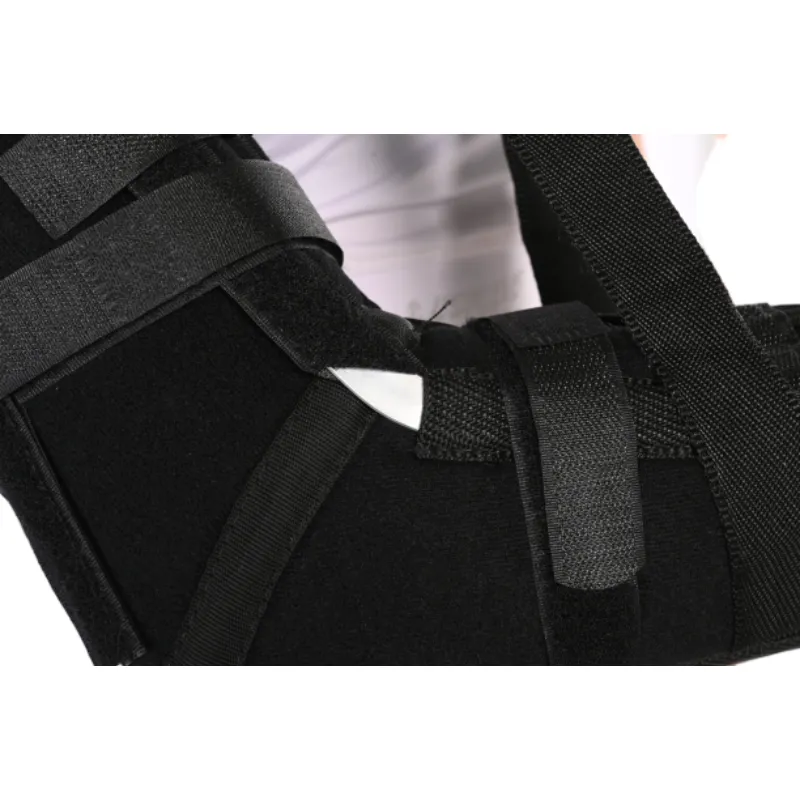
Factory origin: No.240 Xingying West Street, Anping County, Hebei Province, China.
Authoritative references
- American Academy of Orthopaedic Surgeons (AAOS). Elbow injury and postoperative care overviews. Accessed 2025.
- ISO 10993-5/-10: Biological evaluation of medical devices—tests for cytotoxicity and irritation/sensitization.
- ISO 13485: Medical devices—Quality management systems—Requirements for regulatory purposes.
- EU MDR 2017/745: Medical Device Regulation—general safety and performance requirements.
- U.S. FDA. Medical Device Regulations and QSR (21 CFR Part 820) for orthopedic soft goods (general guidance).
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















