Elbow Immobilizer Brace – Medical Support, Adjustable Comfort
Elbow Immobilizer: a field report from clinics, factories, and rehab rooms
If you’re scanning the market for an elbow immobilizer brace, here’s the straight talk. I’ve toured plants, poked at stitch lines, and listened to surgeons and PTs argue over strap geometry. The Elbow Immobilizer from JH Orthopedic (origin: No.240 Xingying West Street, Anping County, Hebei Province, China) keeps coming up in procurement calls—partly because it’s practical, partly because it’s actually built for the messy middle of recovery, not just the catalog photo.

What’s happening in the brace industry
Trends first. Clinicians want breathable spacer fabrics that don’t irritate skin on hour eight. Buyers want consistent sizing and faster lead times. And there’s a quiet shift toward validated materials—biocompatibility, abrasion resistance, and strap life-cycle tests—because returns and skin issues are expensive. Honestly, it’s about comfort plus compliance. If patients actually wear the device, outcomes improve.

Technical snapshot and real-world specs
The Elbow Immobilizer is designed for post-op protection, night positioning, and activity restriction when tendons or the ulnar nerve need peace and quiet. It’s not flashy. It’s a workhorse. Many customers say the straps “just hold” without digging in—small thing, big difference at 3 a.m.
| Feature | Specification (≈, real-world use may vary) |
|---|---|
| Core materials | 6061 aluminum stays + EVA foam + 3D spacer mesh (latex-free) |
| Closure | Hook-and-loop straps, rounded edges; 5,000-cycle closure endurance (internal test) |
| Sizing | S/M/L/XL; biceps circumference ≈ 20–42 cm coverage |
| Positioning | Rigid support to limit elbow flexion/extension; commonly set at functional 90° |
| Weight | ≈ 180–230 g depending on size |
| Service life | Home use ≈ 6–12 months; facility reuse subject to cleaning protocol |
| Testing | Abrasion per ASTM D4966; strap tensile per ASTM D5034; biocompatibility guided by ISO 10993 |

Manufacturing flow, briefly
Materials arrive inspected (fabric GSM, colorfastness, lot traceability). Patterns are CNC-cut for accuracy; stays are deburred and preformed. Panels are stitched using flatlock methods where skin contact is high; edges are bound to reduce rub points. QC includes strap peel strength checks, dimensional tolerance, and random wear-tests. Finished devices are cleaned, bagged with IFU, and boxed. Facilities targeting ISO 13485 typically maintain device history records—yes, paperwork matters.
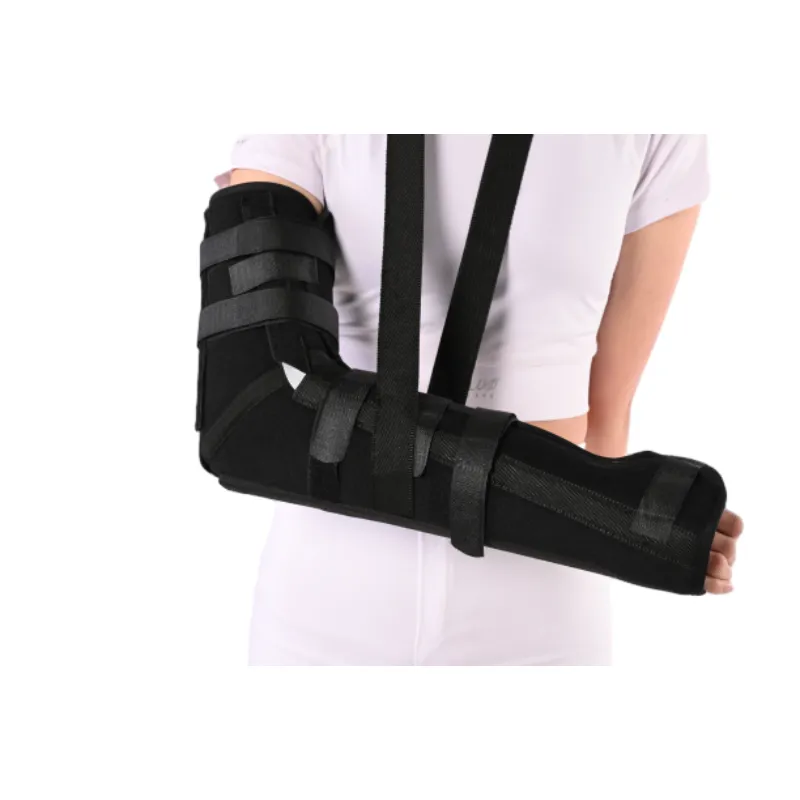
Use cases and advantages
- Post-op tendon repair and ulnar nerve transposition—limit motion, protect sutures.
- Night splinting for cubital tunnel symptoms; to be honest, compliance is the whole game.
- Sports medicine: short-term stabilization post-strain when hinged ROM isn’t prescribed.
- Home rehab and DME supply: straightforward fit, low learning curve.
Advantages? Stable immobilization, breathable contact layer, and a strap layout that doesn’t feel like a tourniquet. Several therapists told me edema control was “easier than expected,” which, surprisingly, you don’t hear often with rigid supports.
Vendor landscape at a glance
| Vendor | Certs (claim) | MOQ | Lead Time | Customization |
|---|---|---|---|---|
| JH Orthopedic | ISO 13485, CE MDR (declared) | ≈ 200 pcs | 15–30 days | Logo, colors, sizes, fabric options |
| Generic Importer A | Basic factory QA | ≈ 500 pcs | 30–45 days | Limited (label only) |
| Premium Brand B | ISO 13485, FDA listing | ≈ 50–100 pcs | 7–20 days | Broad; higher cost |
It seems the sweet spot is certified manufacturing with modest MOQs and sane lead times. For bulk hospital procurement, ask for the technical file and test reports—no shortcuts.
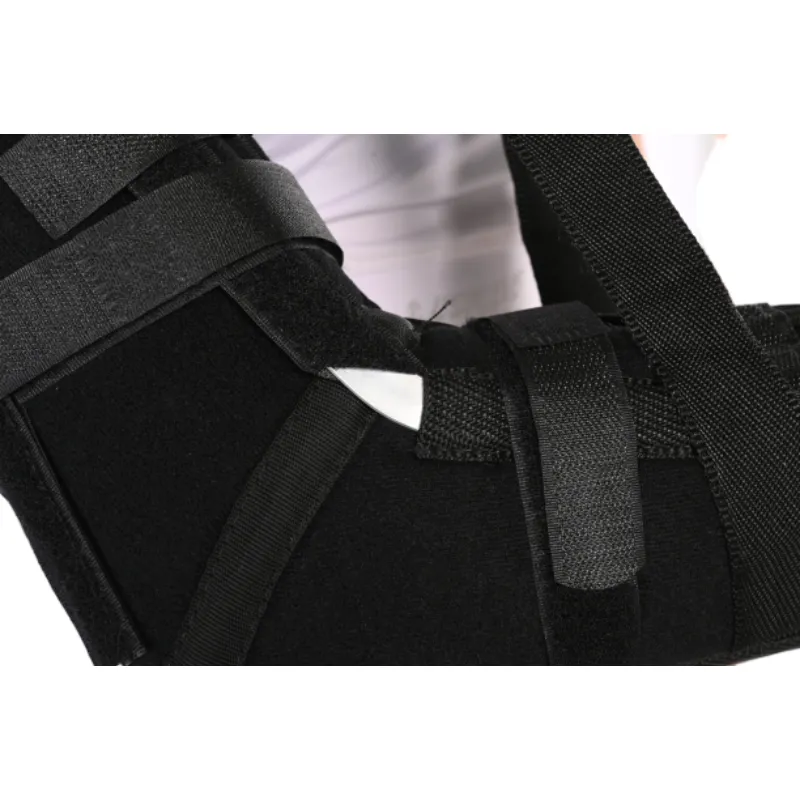
Customization and compliance
Options include antimicrobial liners, extended sizes, pediatric cuts, and OEM branding. For regulatory peace of mind: look for ISO 13485 QMS, EU MDR conformity assessment, and U.S. FDA device listing under Class I (where applicable). Keep cleaning instructions handy; service life depends on reprocessing.
Mini case notes
- A regional ortho clinic reported fewer night-time dropouts after switching to a elbow immobilizer brace with wider forearm straps—comfort drove adherence.
- An e-commerce DME seller cut returns by ≈12% quarter-over-quarter after adding size guides and a elbow immobilizer brace fit video. Small tweaks, big outcome.
References
- ISO 13485:2016 Medical devices—Quality management systems. https://www.iso.org/standard/59752.html
- FDA 21 CFR Part 820—Quality System Regulation. https://www.ecfr.gov/current/title-21/chapter-I/subchapter-H/part-820
- ISO 10993-1: Biocompatibility evaluation. https://www.iso.org/standard/68936.html
- ASTM D4966—Abrasion Resistance of Textile Fabrics (Martindale). https://www.astm.org/d4966
- EU MDR 2017/745—Medical Device Regulation. https://health.ec.europa.eu/medical-devices-sector/new-regulations_en
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















