Neoprene Shoulder Brace: Support, Stability & Pain Relief
Industry Overview and Technical Significance of Neoprene Shoulder Support
The orthopedic and sports medicine sectors continually seek advanced solutions for injury recovery and prevention. Among these, the neoprene shoulder brace stands out as a critical component for therapeutic support. This device, often referred to interchangeably as a neoprene shoulder sleeve or neoprene shoulder stabilizer, is engineered to provide dynamic compression, thermal retention, and proprioceptive feedback to the glenohumeral joint and surrounding musculature. Its efficacy is rooted in the unique properties of neoprene, a synthetic rubber known for its elasticity, durability, and insulation capabilities.
Market trends indicate a robust demand for non-invasive, comfortable, and effective orthopedic supports. The global orthopedic braces and supports market, valued at approximately USD 4.5 billion in 2022, is projected to grow at a compound annual growth rate (CAGR) of around 6.5% from 2023 to 2030. This growth is driven by an aging population, increasing participation in sports, and a rise in musculoskeletal injuries. Specifically, shoulder injuries account for a significant portion of these, with conditions like rotator cuff tears, AC joint sprains, dislocations, and general instability frequently requiring external stabilization. The adoption of advanced materials and ergonomic designs is paramount in meeting this escalating demand, ensuring that products like the neoprene shoulder brace deliver optimal patient outcomes and enhance quality of life.
From a technical perspective, the design of a neoprene shoulder support must consider biomechanical principles to ensure effective stabilization without unduly restricting necessary range of motion. Key design parameters include material thickness, compression zones, strap configurations for adjustable tension, and anatomical contouring to conform seamlessly to the shoulder's complex structure. Manufacturers must adhere to stringent quality control and regulatory standards to guarantee product safety and performance, ensuring that each orthopedic shoulder belt provides consistent therapeutic benefits.
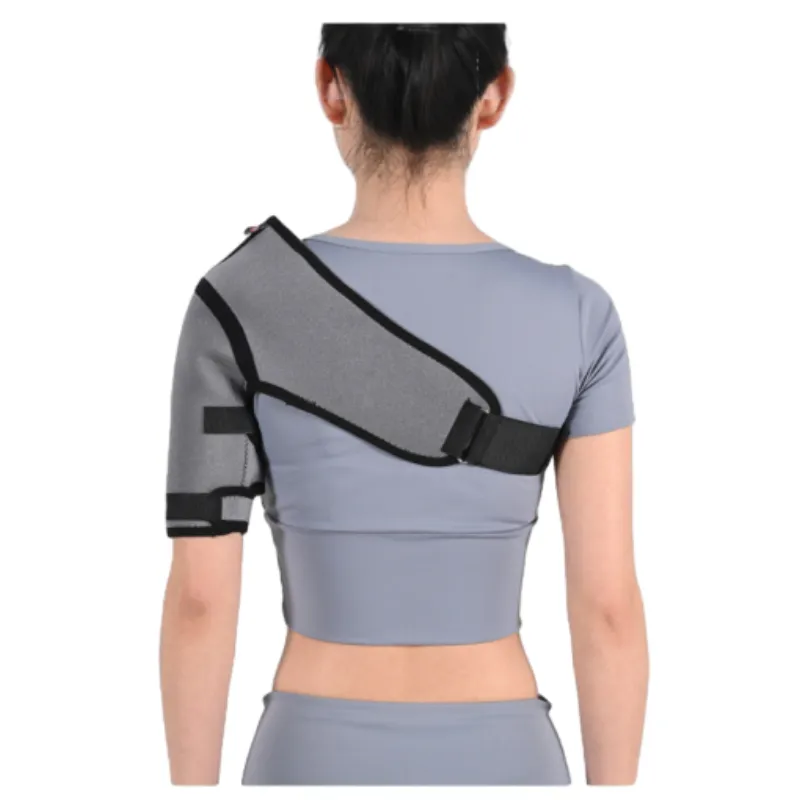
Manufacturing Process of Neoprene Shoulder Braces
The production of a high-quality neoprene shoulder brace is a multi-stage process, demanding precision engineering and adherence to medical device manufacturing protocols. This ensures durability, comfort, and therapeutic efficacy.
- Material Sourcing and Preparation: The foundational material is medical-grade neoprene, typically a closed-cell foam laminated with a stretchable nylon or polyester fabric on one or both sides. This lamination enhances durability, provides a smooth finish, and prevents direct skin contact with the rubber. Materials are rigorously tested for biocompatibility (ISO 10993), skin irritation, and allergenicity, often complying with REACH and RoHS directives for chemical safety.
- Precision Pattern Cutting: CAD (Computer-Aided Design) software is used to generate precise patterns for the various components of the neoprene shoulder sleeve. These digital patterns are then fed into automated cutting machines, such as CNC laser cutters or die-cutting presses. This ensures high dimensional accuracy and consistency across batches, critical for effective anatomical fit and compression.
- Component Assembly and Stitching: Individual cut pieces are assembled and stitched together. Flatlock stitching is commonly employed to create smooth seams that lie flat against the skin, minimizing irritation and pressure points, which is crucial for a comfortable neoprene shoulder stabilizer intended for prolonged wear. High-strength nylon or polyester threads are used, selected for their durability and resistance to stretching. Reinforced stitching is applied at stress points, such as where straps attach or at major seams, to prevent tearing.
- Fastener Integration: High-quality hook-and-loop fasteners (e.g., Velcro-brand equivalent) are sewn onto the brace. The placement and type of fastener are critical for allowing users to adjust the compression level and fit, accommodating various body types and injury-specific requirements. The durability of these fasteners is tested for thousands of cycles to ensure long-term functionality.
- Finishing and Quality Control (QC): After assembly, each orthopedic shoulder belt undergoes a thorough inspection. This includes checks for stitching integrity, material flaws, correct sizing, fastener functionality, and overall aesthetic quality. Dimensional accuracy is verified using specialized gauges. Testing standards often include ISO 13485 (Quality Management System for Medical Devices), ensuring a documented and controlled manufacturing environment. Performance testing may involve simulated wear cycles to assess durability and compression retention.
- Packaging and Sterilization (if applicable): Braces are cleaned, folded, and packaged, often in moisture-resistant bags to maintain hygiene. While neoprene braces are typically non-sterile devices, certain medical applications might require specific sterilization protocols or packaging in cleanroom environments, although this is less common for standard OTC shoulder braces.
Advantages in Application Scenarios:
- Enhanced Recovery: The thermogenic properties of neoprene increase local blood circulation, promoting healing for conditions like tendinitis or muscle strains.
- Superior Stabilization: Provides proprioceptive feedback, reminding the user of safe movement limits, crucial for post-dislocation or rotator cuff injury recovery.
- Comfort and Durability: Soft, flexible materials ensure patient compliance for long-term wear, while robust construction guarantees an extended service life of typically 1-3 years with proper care.
Technical Specifications and Performance Parameters
Understanding the technical specifications of a neoprene shoulder brace is paramount for B2B decision-makers, ensuring that the chosen product aligns with specific clinical requirements and patient demographics. Below is a representative table of critical parameters for the "Neopere Shoulder Support".
Product Specification Table: Neopere Shoulder Support
| Parameter | Specification | Technical Standard / Compliance |
|---|---|---|
| Material Composition | 70% Neoprene, 20% Nylon, 10% Polyester (Lycra blend for elasticity) | ISO 10993 (Biocompatibility), REACH, RoHS |
| Neoprene Thickness | 3.0 mm (optimal for compression and flexibility) | +/- 0.2 mm manufacturing tolerance |
| Compression Level | Moderate (15-25 mmHg at rest, adjustable) | ASTM F1725-09 (Standard Specification for Therapeutic Compression Devices) |
| Sizing Availability | S, M, L, XL (based on chest circumference: S: 28-34", M: 35-40", L: 41-46", XL: 47-52") | Anthropometric data derived from ISO 7250 |
| Fastener Type | High-cycle hook-and-loop (e.g., Velcro-equivalent) | Tested for >10,000 cycles without significant loss of adhesion |
| Care Instructions | Hand wash cold, air dry. Do not bleach or iron. | Ensures longevity and material integrity |
| Intended Use | Rotator cuff support, AC joint sprain, post-surgical recovery, muscle strains, general shoulder instability | Clinical indication per FDA 21 CFR Part 880 (Medical Devices) |
These detailed specifications ensure that medical professionals and distributors can confidently select the appropriate neoprene shoulder support for their clients, knowing it meets rigorous performance and safety criteria. The combination of material science and ergonomic design contributes directly to the efficacy and user acceptance of the product.

Application Scenarios and Case Studies
The versatility of the neoprene shoulder brace makes it indispensable across various clinical and active settings. Its primary function is to provide compression, warmth, and stabilization to the shoulder joint, which significantly aids in recovery and injury prevention.
Typical Application Scenarios:
- Post-Surgical Rehabilitation: Following procedures like arthroscopy for rotator cuff repair or labral reconstruction, a neoprene shoulder stabilizer can provide gentle compression and warmth, reducing edema and improving blood flow, thereby accelerating the healing process. It acts as a transitional support as patients progress from immobilization to active rehabilitation.
- Sports Injury Management: Athletes involved in overhead sports (e.g., baseball, tennis, swimming) or contact sports (e.g., rugby, football) frequently experience shoulder strains, sprains, and minor dislocations. A neoprene shoulder sleeve offers prophylactic support during training and competition or aids in the initial stages of recovery for acute injuries.
- Chronic Conditions and Overuse Injuries: Patients suffering from chronic conditions such as osteoarthritis, tendinitis (e.g., bicipital tendinitis), or bursitis benefit from the continuous warmth and compression, which can alleviate pain and reduce inflammation.
- Occupational Health: Workers whose jobs involve repetitive arm movements or heavy lifting, such as in construction or manufacturing, can use an orthopedic shoulder belt to prevent repetitive strain injuries and provide support during daily tasks.
Application Case Studies:
Case Study 1: Rotator Cuff Tendinitis in a Professional Golfer
A 35-year-old professional golfer presented with chronic tendinitis in his dominant shoulder, impacting his swing mechanics and performance. Traditional physical therapy yielded slow progress. His orthopedist prescribed a custom-fitted neoprene shoulder brace for use during daily activities and light training. The brace provided continuous therapeutic warmth and compression, reducing pain and improving local circulation. Within 4 weeks, the patient reported significant pain reduction (from 7/10 to 3/10 on the VAS scale) and increased range of motion, allowing him to resume full practice with reduced risk of re-injury. The proprioceptive feedback also helped him maintain proper shoulder alignment.
Case Study 2: Post-Dislocation Rehabilitation in an Adolescent Athlete
A 17-year-old high school basketball player experienced a traumatic anterior shoulder dislocation. After successful reduction and initial sling immobilization, a neoprene shoulder support was integrated into his rehabilitation protocol. The brace offered crucial support during guided exercises, providing a sense of security and preventing excessive external rotation, which is a common mechanism for re-dislocation. The patient showed accelerated progress in regaining strength and stability, returning to non-contact sports after 8 weeks and full participation within 12 weeks, significantly faster than typical recovery times often cited at 16-20 weeks without such targeted support.

Technical Advantages and Vendor Comparison
The selection of a neoprene shoulder brace often involves a detailed evaluation of technical advantages and a comparison between various vendors. Our "Neopere Shoulder Support" offers distinct benefits.
Key Technical Advantages of Neopere Shoulder Support:
- Advanced Neoprene Formulation: Utilizes a premium, breathable perforated neoprene, which mitigates heat buildup and moisture accumulation, a common issue with traditional neoprene, enhancing comfort during extended wear.
- Ergonomic & Biomechanical Design: Engineered with a dynamic anatomical fit that contours precisely to the shoulder joint, optimizing compression distribution and providing superior proprioceptive feedback without restricting essential range of motion for rehabilitation.
- Reinforced Stitching & Durability: Incorporates multi-directional flatlock stitching and reinforced seams at high-stress points, ensuring exceptional durability and resistance to wear and tear, even with rigorous athletic use.
- Micro-Adjustable Compression System: Features a dual-strap adjustment mechanism that allows for precise, individualized compression levels, accommodating different injury types and stages of recovery, promoting targeted support.
- Hypoallergenic Liner: Lined with a soft, moisture-wicking fabric that is gentle on the skin, reducing the risk of irritation and allergic reactions, suitable for sensitive skin types.
Vendor Comparison: Neopere Shoulder Support vs. Generic Neoprene Braces
| Feature/Criterion | Neopere Shoulder Support | Generic Neoprene Brace |
|---|---|---|
| Material Quality | Premium perforated neoprene, breathable, medical-grade, ISO 10993 certified. | Standard neoprene, often non-perforated, lower-grade, variable certifications. |
| Design & Ergonomics | Anatomically contoured, low-profile, supports full range of motion. | Often bulky, less specific contouring, can restrict natural movement. |
| Compression Adjustment | Dual-strap system for precise, micro-adjustable compression zones. | Single strap or limited adjustment, less targeted compression. |
| Durability & Stitching | Flatlock stitching, reinforced seams, high-cycle fasteners (>10,000 uses). | Standard stitching, prone to fraying, fasteners degrade quickly. |
| Patient Comfort | Breathable, hypoallergenic liner, minimal skin irritation, long-term wear comfort. | Can cause sweating, skin irritation, uncomfortable for prolonged use. |
| Certifications | ISO 13485, CE Marking, FDA Registered (Product-specific) | Often lacks comprehensive medical device certifications. |
Customized Solutions and Partner Integration
Recognizing the diverse needs of medical distributors, sports organizations, and healthcare providers, we offer extensive customized solutions for our neoprene shoulder brace product line. This flexibility extends beyond standard sizing to bespoke branding and functional modifications, ensuring seamless integration into varied operational models.
Customization Options:
- Branding and Packaging: Private labeling, custom logos, and branded packaging solutions are available. This allows partners to market the neoprene shoulder sleeve under their own brand identity, strengthening their market presence.
- Material Modifications: Custom requests for specific neoprene thicknesses (e.g., 2.5mm for lighter support, 4.0mm for enhanced compression), different fabric laminates (e.g., antimicrobial treatments, higher wicking properties), or color variations can be accommodated for bulk orders.
- Design Adaptations: For unique clinical requirements, minor design modifications such as additional strap configurations, integrated gel pads for targeted compression, or variations in anatomical cut can be implemented. Our R&D team works closely with partners to develop prototypes and ensure design efficacy.
- Sizing and Fit: Beyond standard S-XL, we offer options for pediatric or bariatric sizing, and we can develop size charts tailored to specific regional anthropometric data for markets outside standard Western demographics.
Our strategic partnerships include leading orthopedic clinics, major sports equipment retailers, and national rehabilitation centers. These collaborations underscore our commitment to providing tailored solutions that meet the stringent demands of diverse B2B clients, ensuring the orthopedic shoulder belt we supply is optimally suited for its intended application and user base.
Trustworthiness and Support: FAQ, Lead Time, Warranty
Building trust with our B2B partners is paramount. We achieve this through transparent processes, robust quality assurance, and comprehensive customer support for every neoprene shoulder brace we manufacture.
Frequently Asked Questions (FAQ):
- Q1: What certifications do your neoprene shoulder braces hold?
- A1: Our manufacturing facilities are ISO 13485 certified, and our products carry CE Marking, signifying compliance with European health, safety, and environmental protection standards. We also adhere to FDA regulations for medical devices in relevant markets.
- Q2: What is the typical lead time for bulk orders of the Neopere Shoulder Support?
- A2: Standard lead time for bulk orders (typically 500+ units) is 4-6 weeks from order confirmation and deposit. For customized solutions or larger volumes, lead times may extend to 8-10 weeks, depending on the complexity of the customization and material availability. Expedited options may be available upon request and with a premium.
- Q3: What is your warranty policy for the neoprene shoulder brace?
- A3: We offer a 1-year limited warranty against manufacturing defects in materials and workmanship from the date of purchase. This warranty covers issues arising from normal use. It does not cover damage caused by improper use, accident, abuse, or unauthorized modifications. Specific details are provided in our official warranty statement.
- Q4: How do you ensure product quality and consistency for large orders?
- A4: We implement a rigorous three-stage quality control process: Incoming Quality Control (IQC) for raw materials, In-Process Quality Control (IPQC) throughout manufacturing, and Final Quality Control (FQC) for finished goods. Each batch undergoes sampling and comprehensive testing to ensure adherence to specified parameters and standards, minimizing defect rates to less than 0.5%.
After-Sales Support:
Our commitment extends beyond delivery. We provide dedicated after-sales support, including technical assistance, troubleshooting guides, and a streamlined process for warranty claims or product returns. Our customer service team is available via phone and email during business hours to assist our B2B partners, ensuring a seamless experience and continued satisfaction with our neoprene shoulder stabilizer products. We also offer training materials and product information sessions for distributor sales teams to enhance their understanding and selling capabilities.
References
- Grand View Research. (2023). Orthopedic Braces And Support Systems Market Size, Share & Trends Analysis Report By Product, By Application, By End-Use, By Region, And Segment Forecasts, 2023 - 2030.
- International Organization for Standardization. (2016). ISO 13485:2016 Medical devices — Quality management systems — Requirements for regulatory purposes.
- U.S. Food and Drug Administration. (2023). Medical Devices.
- American Society for Testing and Materials. (2009). ASTM F1725-09 Standard Specification for Therapeutic Compression Devices.
- European Chemicals Agency. (2023). REACH – Registration, Evaluation, Authorisation and Restriction of Chemicals.
-
Thumb Spica Belt Guide for Thumb Stabilization and RecoveryNews Apr.16,2026
-
Soft Thumb Spica Guide for Effective Thumb Stabilization and HealingNews Apr.09,2026
-
Comprehensive Guide to Hand Spica Casts for Effective Injury RecoveryNews Apr.07,2026
-
Benefits of a Custom Thumb Spica for Optimal Hand RecoveryNews Apr.04,2026
-
Experience Ultimate Comfort Cool Thumb Spica for Wrist and Thumb SupportNews Mar.31,2026
-
Comprehensive Guide to Choosing the Right Youth Thumb Splint for Effective HealingNews Mar.28,2026





















