Pouch Arm Sling with Immobilizer – Adjustable, Breathable
Field Notes on the Modern pouch arm sling with immobilizer: What Clinicians and Buyers Should Know
I’ve toured a few brace factories in my time, and—surprisingly—the quiet star of post-op shoulder care isn’t flashy tech; it’s fit, fabric, and the little reinforcements you can’t see. The Arm Sling Pouch coming out of No.240 Xingying West Street, Anping County, Hebei Province, China, strikes that balance. To be honest, many customers say it just “disappears” once fitted right—exactly what you want after a rotator cuff repair or a stubborn dislocation.

Industry trend: breathable, lighter, yet tougher
Clinics are moving toward breathable spacer-mesh pouches and detachable bolsters that hold the humerus stable without cooking the skin. Procurement teams want two things: fast adjustability in the ER and reliable immobilization for the first 6–12 weeks. The pouch arm sling with immobilizer format ticks both boxes—and yes, it still needs industrial-grade stitching where the strap meets the pouch. That’s where cheaper models often fail.
Technical specifications (typical)
| Parameter | Spec (≈/typical) | Notes (real-world use may vary) |
|---|---|---|
| Pouch fabric | 3D spacer mesh + brushed polycotton lining | Breathable, soft hand-feel |
| Immobilizer bolster | Detachable foam, 35–45 kg/m³ | Maintains abduction angle |
| Straps & closure | 38 mm nylon webbing + hook-and-loop (5 cm) | Bartack reinforced, padded shoulder |
| Adjustability | ≈140–200 cm circumference | Universal R/L; one-hand tightening |
| Breathability | Air permeability ≈250 mm/s (ASTM D737) | Reduced skin maceration risk |
| Load & durability | System tensile >600 N; 5,000+ VELCRO cycles | ASTM D5034 / D5169 reference |
| Care life | ≈30 washes @40°C; latex-free | Clinic re-use depends on SOPs |
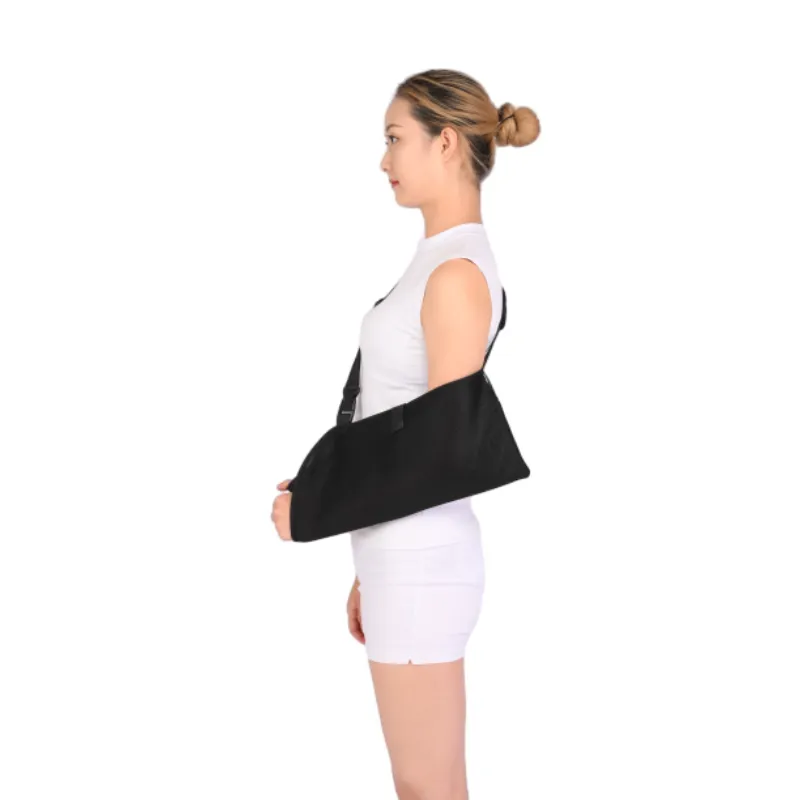
How it’s made (process flow)
- Materials: OEKO-TEX-style fabrics, medical-grade foam, nylon webbing, low-allergen hook-and-loop.
- Cutting & sewing: CNC cutting, ultrasonic edge sealing, 8–12 bartacks at stress points.
- Bolster shaping: density-matched foam for stable abduction without pressure hot-spots.
- Testing: tensile (ASTM D5034), hook-and-loop shear (ASTM D5169), air permeability (ASTM D737), biocompatibility screening (ISO 10993-5).
- Service life: typically 6–12 weeks patient use; rental fleets ≈12 months with validated cleaning.
- Industries: hospitals, ambulatory surgery centers, sports med, EDs, DME retailers, physio clinics.

Real-world use cases
Post–rotator cuff repair, capsular shift, clavicle fractures, acute dislocations, even long flights where motion control matters. Clinicians like the quick height adjustment; patients like that the pouch arm sling with immobilizer doesn’t feel swampy after an hour. I guess that’s the spacer mesh earning its keep.
Vendor snapshot (at a glance)
| Vendor | Certs | Lead time | Customization | Notes |
|---|---|---|---|---|
| JH Orthopedic (China) | ISO 13485; CE MDR Class I; FDA Class I (880.6300) | ≈15–30 days | Logo, colors, pediatric sizing, packaging | Factory-direct; competitive MOQs |
| EU Brand M | ISO 13485; CE MDR | ≈30–45 days | Moderate | Higher unit cost, strong distributor support |
| US Brand S | FDA listed; ISO 13485 | Stock-dependent | Limited | Fast domestic replenishment |
Testing data and feedback
- Strap-to-pouch tensile: >600 N; creep
- Hook-and-loop retention: ≥85% shear strength after 5,000 cycles (ASTM D5169).
- ISO 10993-5 cytotoxicity: non-cytotoxic response reported by manufacturer.
- Customer notes: “shoulder pad actually helps on long wear”; “bolster stays put during PT transfers.”

Customization playbook
Private label? Easy. Typical options: brand tags, dye-matched webbing, QR-coded IFU, pediatric silhouettes, antimicrobial finishes, and bilingual packaging. For tenders, request documented ISO 13485 scope, MDR Class I declaration, and FDA product listing. It seems that the pouch arm sling with immobilizer is now as much about paperwork as padding.
Quick application tips
- Fit elbow deep in the pouch; adjust sling height so wrist is level with elbow.
- Seat the bolster before final strap tensioning to avoid neck load.
- Recheck after 15 minutes—micro-adjustments reduce pressure points.
References
- ISO 13485:2016 — Medical devices — Quality management systems.
- FDA 21 CFR 880.6300 — Arm sling; Class I, generally exempt.
- ISO 10993-5 — Biological evaluation of medical devices — Tests for in vitro cytotoxicity.
- ASTM D5034 — Standard Test Method for Breaking Strength and Elongation of Textile Fabrics (Grab Test).
- Regulation (EU) 2017/745 — Medical Device Regulation (MDR), Class I.
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















