Secure Pouch Arm Sling with Immobilizer for Optimal Recovery
Industry Trends in Orthopedic Immobilization Devices
The global market for orthopedic immobilization devices is experiencing robust growth, driven by an aging population, increasing incidence of sports- related injuries, and advancements in non-invasive treatment modalities. A significant trend observed is the demand for patient-centric solutions that balance effective immobilization with comfort and ease of use. This shift is propelling innovation in products like the pouch arm sling with immobilizer, which offers superior support and reduces recovery times. Technological integration, such as breathable fabrics, ergonomic designs, and modular components, is paramount. Furthermore, the emphasis on evidence-based rehabilitation protocols necessitates devices that provide consistent, measurable support, leading to a higher demand for scientifically engineered slings and braces.
Key market drivers include the rising prevalence of musculoskeletal disorders, especially those affecting the shoulder and upper extremities, and the increasing adoption of minimally invasive surgical techniques that still require post-operative support. The market also sees a growing preference for lightweight, durable, and hypoallergenic materials, which directly influences product development and manufacturing processes. Providers are increasingly seeking solutions that are cost-effective for bulk procurement while maintaining high standards of clinical efficacy and patient satisfaction.

Manufacturing Process: Precision Engineering for Orthopedic Support
The production of a high-quality pouch arm sling with immobilizer involves a meticulous multi-stage manufacturing process, ensuring both clinical efficacy and patient comfort. Unlike heavy industrial components, the focus here is on advanced textile engineering and ergonomic design principles. Our process adheres strictly to ISO 13485 quality management systems for medical devices, guaranteeing consistency and reliability.
Process Flow Schematic:
- Material Sourcing & Quality Inspection: Procurement of medical-grade, breathable fabrics (e.g., polyester, nylon blends), high-density foam padding for comfort, durable polypropylene webbing for straps, and strong hook-and-loop fasteners. All raw materials undergo stringent incoming quality control for tensile strength, skin compatibility, and compliance with ISO and FDA standards.
- Precision Cutting: Automated CNC fabric cutting machines are used to precisely cut pouch components, shoulder straps, and immobilizer bands according to predefined CAD designs. This ensures dimensional accuracy and minimizes material waste.
- Stitching & Assembly: Skilled technicians utilize industrial sewing machines for durable, reinforced stitching. Critical areas, such as D-ring attachments and strap junctions, receive extra reinforcement to withstand continuous stress. Foam padding is integrated into the shoulder strap for comfort, and the thumb loop is securely attached within the pouch.
- Immobilizer Integration: The specialized immobilizer strap, often an adjustable waist strap, is integrated to prevent external and internal rotation, and abduction, ensuring comprehensive shoulder immobilization. This requires precise alignment and robust attachment points.
- Quality Assurance & Testing: Each finished sling undergoes a multi-point inspection. This includes checks for stitching integrity, fastener strength, dimensional conformity, and overall functionality. Batch testing may include stress tests on straps and fasteners, and material durability assessments. Products must meet international standards such as CE marking for European markets and FDA clearance for the US.
- Packaging & Sterilization (Optional): Slings are carefully folded and packaged, often with individual instructions for use. For specific medical environments, a sterilization process may be applied, though typically these devices are not sterile by default but rather cleaned for medical use.
The service life of a well-manufactured shoulder arm pouch is typically 1-3 years under normal use, though this can vary based on patient activity levels and adherence to care instructions. These devices are crucial in target industries such as hospitals (orthopedic, emergency departments), rehabilitation centers, sports medicine clinics, and primary care facilities. Advantages in typical application scenarios include enhanced patient compliance due to comfort, optimal healing environment through stable immobilization, and a significant reduction in post-operative complications related to unintended movement. The superior design of the shoulder sling pouch promotes rapid recovery and improved patient outcomes.
Technical Specifications and Parameters
Understanding the precise technical parameters of a sling arm pouch is critical for healthcare professionals to select the most appropriate device for specific clinical needs. Our products are engineered with robust specifications to ensure superior patient care and durability.
Typical Product Specifications:
| Parameter | Specification | Details / Standard |
|---|---|---|
| Material Composition (Pouch) | Polyester-Nylon Blend | Breathable, moisture-wicking, hypoallergenic. |
| Material Composition (Straps) | Polypropylene Webbing, High-Density Foam | Durable, non-stretch, pressure distribution. |
| Immobilizer Type | Integrated Waist/Abduction Strap | Prevents rotation and unwanted abduction. |
| Adjustability | Fully Adjustable (Shoulder, Waist, Thumb Loop) | Accommodates various body types and injury positions. |
| Sizes Available | S, M, L, XL (based on forearm length) | Refer to sizing chart for specific measurements. |
| Weight | ~0.25 - 0.45 kg (size dependent) | Lightweight for patient comfort. |
| Certifications | ISO 13485, CE, FDA Registered | Ensures international quality and safety standards. |
| Cleaning Instructions | Hand wash cold, air dry | Maintains material integrity and hygiene. |
These specifications highlight our commitment to providing an advanced arm shoulder sling support that meets the rigorous demands of professional medical environments.
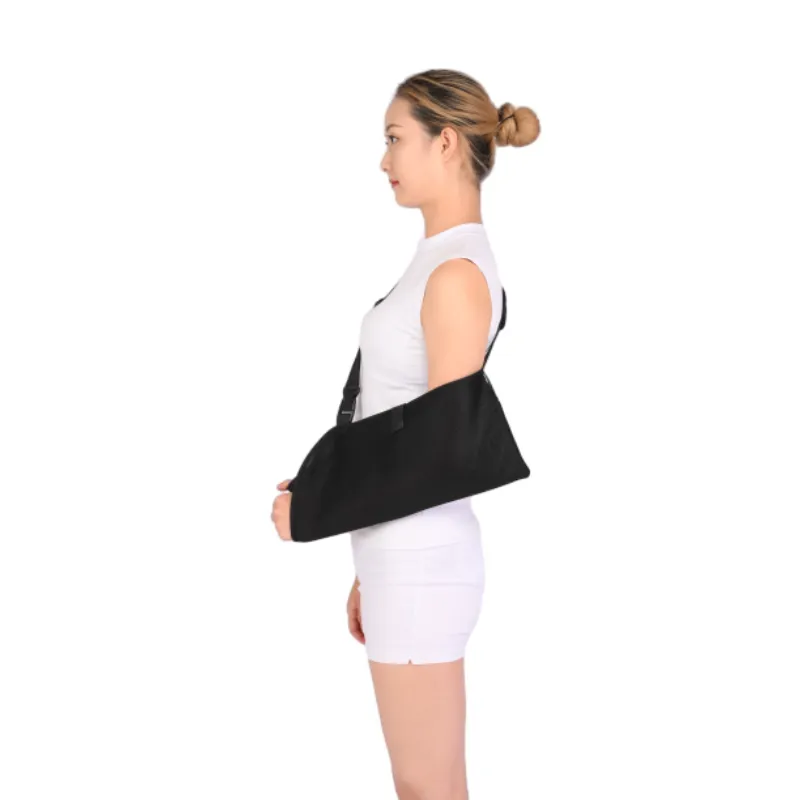
Application Scenarios and Technical Advantages
The versatility and robust design of the pouch arm sling with immobilizer make it indispensable across a range of medical and rehabilitative settings. Its primary function is to provide stable and comfortable support to the arm and shoulder, facilitating optimal healing and pain management.
Key Application Scenarios:
- Post-Operative Recovery: Essential after rotator cuff repairs, shoulder arthroscopy, labral repairs, and other reconstructive shoulder surgeries. The immobilizer ensures the arm remains in a safe, non-stressful position, preventing accidental movements that could compromise surgical outcomes.
- Fracture Immobilization: Used for stable fractures of the humerus, clavicle, scapula, and certain forearm fractures. It provides consistent support, crucial for bone union and reducing displacement.
- Sprains and Strains: Offers necessary rest and support for severe sprains of the shoulder, elbow, or wrist, and muscle strains in the upper arm or deltoid region.
- Shoulder Dislocations/Subluxations: Helps to stabilize the shoulder joint after reduction, minimizing the risk of recurrent dislocation during the healing phase.
- Neurological Conditions: Can be used to support a paretic or flaccid arm in patients with neurological deficits (e.g., stroke), preventing shoulder subluxation and related pain.
Technical Advantages:
- Enhanced Immobilization: The integrated waist/abduction strap effectively limits external rotation and abduction, preventing unwanted movement of the shoulder joint far more effectively than a standard sling. This is critical for specific surgical protocols.
- Patient Comfort and Compliance: Constructed with breathable, moisture-wicking materials and padded straps to reduce skin irritation, heat buildup, and pressure points, leading to higher patient compliance over extended wear periods.
- Ergonomic Design: Contoured pouch and adjustable components ensure a precise fit for various arm lengths and body types, promoting anatomical alignment and reducing strain on the neck and contralateral shoulder.
- Durability and Longevity: Reinforced stitching and high-quality fasteners (hook-and-loop) ensure the product withstands daily wear and multiple adjustments, crucial for the intensive demands of rehabilitation.
- Ease of Application and Adjustment: Designed for intuitive application by medical staff and patients, featuring quick-release buckles and easily adjustable straps for tailored support and minimal disruption during examinations or personal hygiene.
- Hygiene and Maintenance: Materials are selected for easy cleaning, supporting infection control protocols in clinical environments.
These technical advantages position our arm shoulder sling support as a preferred choice for institutions seeking reliable, patient-friendly, and clinically effective orthopedic solutions.
Vendor Comparison and Customized Solutions
In a competitive market for orthopedic devices, choosing the right vendor for a pouch arm sling with immobilizer is critical. Factors such as product quality, customization capabilities, and post-sales support significantly impact patient outcomes and institutional efficiency. We differentiate ourselves through a commitment to superior engineering and comprehensive client service.
Vendor Comparison Overview:
| Feature/Service | Our Offering (JHorpedic) | Standard Industry Offerings |
|---|---|---|
| Material Quality | Premium, medical-grade, breathable, hypoallergenic fabrics and durable fasteners. ISO 13485 certified. | Varies widely; can include less breathable or lower durability materials. |
| Immobilization Efficacy | Integrated, multi-point immobilization (e.g., thumb loop, waist strap) for comprehensive stability. | Often basic slings with limited or add-on immobilization features. |
| Customization Options | Extensive (branding, sizing, material, specific design modifications for bulk orders). | Limited to standard sizing and colors, minimal design flexibility. |
| Quality Control & Certifications | Rigorous multi-stage QA, ISO 13485, CE, FDA registration. | May vary, some may lack specific medical device certifications. |
| Lead Times & Fulfillment | Streamlined logistics, competitive lead times, global distribution. | Can be inconsistent, particularly for specialized orders. |
| Customer Support | Dedicated B2B support, technical assistance, warranty. | Often generic, less specialized support. |
Customized Solutions:
Recognizing that unique clinical requirements demand tailored solutions, we offer extensive customization options for our shoulder arm pouch for bulk institutional orders. This flexibility ensures that our partners receive products perfectly aligned with their specific patient demographics and operational needs:
- Size Modifications: Custom sizing for pediatric patients, bariatric individuals, or populations with atypical limb proportions.
- Material Adjustments: Option for specialized antimicrobial fabrics, enhanced breathability for tropical climates, or specific hypoallergenic materials for sensitive skin.
- Branding and Packaging: Private labeling, custom branding, or specific packaging configurations for ease of distribution within large healthcare networks.
- Feature Integration: Addition of specialized pockets, alternative closure mechanisms, or design modifications to accommodate specific medical devices or patient needs.
Our engineering team collaborates closely with clients from conceptualization to delivery, ensuring that customized orders meet stringent performance and quality benchmarks.

Application Case Studies and Testimonials
Real-world application demonstrates the tangible benefits of our pouch arm sling with immobilizer. Our long-standing relationships with leading healthcare providers globally attest to the product's effectiveness and reliability in diverse clinical scenarios.
Case Study 1: Post-Rotator Cuff Repair Rehabilitation
Client: Major Metropolitan Hospital, Orthopedic Department
Challenge: The hospital experienced patient non-compliance with post-operative immobilization protocols following rotator cuff repair, leading to a higher incidence of re-ruptures and prolonged rehabilitation. Standard slings were found to be uncomfortable and offered insufficient rotational stability.
Solution: The hospital integrated our shoulder sling pouch with an integrated abduction pillow and immobilizer strap as the standard post-operative device. The enhanced comfort features, combined with superior immobilization, significantly improved patient adherence.
Outcome: Over a 12-month period, the hospital reported a 30% reduction in re-rupture rates among patients using our sling, alongside an average 15% reduction in overall rehabilitation time. Patient satisfaction surveys indicated a significant increase in perceived comfort and ease of use.
Case Study 2: Sports Injury Management at a University Clinic
Client: Regional University Sports Medicine Clinic
Challenge: The clinic required a durable and highly effective arm immobilizer for athletes recovering from shoulder dislocations and acromioclavicular (AC) joint separations. The devices needed to withstand active lifestyles (within prescribed limits) and facilitate a quick return to sport-specific rehabilitation.
Solution: The clinic adopted our specialized sling arm pouch, customized with reinforced stitching and quick-dry materials, providing athletes with robust support while allowing for greater hygiene during their recovery. The adjustable nature ensured precise fit for varying body types of student-athletes.
Outcome: The clinic observed improved early-stage healing and reduced pain scores among athletes. The product's durability proved critical in preventing premature wear and tear, ensuring consistent support throughout the initial immobilization phase. The ability to quickly adjust and remove the sling for physical therapy sessions was also highly valued by both clinicians and athletes.

Trustworthiness: FAQ, Lead Time, Warranty, and Support
At JHorpedic, building trust with our B2B partners is paramount. We achieve this through transparent operations, clear commitments, and robust support systems for every arm shoulder sling support we provide.
Frequently Asked Questions (FAQ):
Q: What materials are used in your pouch arm sling with immobilizer?
A: Our slings are primarily constructed from a durable, breathable polyester-nylon blend for the pouch, with high-density foam padding and robust polypropylene webbing for the straps. All materials are selected for patient comfort, durability, and skin compatibility, adhering to medical device standards.
Q: Are your products certified for medical use?
A: Yes, all our orthopedic immobilization devices, including the shoulder arm pouch, are manufactured under ISO 13485 certified quality management systems. They also hold CE marking for the European market and are FDA registered for sale in the United States, affirming their compliance with international safety and quality standards.
Q: How do I determine the correct size for a patient?
A: We provide detailed sizing charts based on forearm length and chest circumference. For optimal fit, measure from the elbow crease to the base of the little finger. Our customer support team can also assist with specific sizing inquiries for bulk orders or custom solutions.
Lead Time & Fulfillment Details:
We understand the critical nature of timely supply in healthcare. Our standard lead time for in-stock bulk orders of the shoulder sling pouch typically ranges from 7 to 14 business days, depending on order volume and destination. For customized solutions, lead times will be provided upon project scope confirmation, factoring in material sourcing and specific manufacturing requirements. We maintain a robust inventory and efficient logistics network to ensure prompt and reliable delivery worldwide.
Warranty Commitments:
JHorpedic products are backed by a 1-year limited warranty against manufacturing defects from the date of purchase. This warranty covers defects in materials and workmanship under normal use and service. It does not cover damage resulting from misuse, alteration, accident, or normal wear and tear. Our commitment to quality ensures that our products meet or exceed industry standards, providing confidence in their performance and durability.
Customer Support Information:
Our dedicated B2B customer support team is available to assist with product inquiries, technical specifications, order placement, and after-sales service. For immediate assistance, please visit our website's contact section or reach out via email at sales@jhorthopedic.com. We pride ourselves on responsive and knowledgeable support to ensure a seamless partnership.
Authoritative References
- International Organization for Standardization. ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes.
- U.S. Food and Drug Administration. Medical Devices.
- American Academy of Orthopaedic Surgeons. Shoulder Injury and Treatment.
- European Commission. Medical Devices Regulation (EU) 2017/745.
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















