Shoulder Brace Support Arm Sling: Adjustable, Fast Recovery?
Shoulder Brace Support Arm Sling: Field Notes from Clinics and Factory Floors
If you spend time in post-op units or sports-medicine clinics, you notice something: a good sling or immobilizer can calm an entire room. Patients settle. Nurses move quicker. And, to be honest, surgeons sleep better. That’s where the [shoulder brace support arm sling] concept behind the Eblow Brace comes in—an upper-limb support engineered for those messy, critical weeks after injury or surgery.

The Eblow Brace (yes, spelled “Eblow”) ships from No.240 Xingying West Street, Anping County, Hebei Province, China, and it behaves like a modular upper-limb system: sling-style shoulder support, elbow stabilization, and forearm positioning in one package. In fact, many customers say it feels more breathable than the old neoprene rigs; surprisingly, that matters on day three when the bandage itch kicks in.
Industry trends and where this fits
We’re seeing three converging trends: lighter spacer-mesh laminates, recyclable aluminum stays, and hospital-grade hook-and-loop that survives disinfection protocols. Hospitals want multi-size kits that cover 80% of patients; home rehab wants easy don/doff and clear strap routing. The Eblow Brace leans into all three, and—actually—does it without the bulky look.
Key specifications (lab and real-world)
| Parameter | Spec (≈, real-world may vary) |
|---|---|
| Materials | 3D spacer mesh + brushed nylon liner; aluminum stays; medical-grade hook-and-loop |
| Adjustability | Forearm cradle 28–38 cm; shoulder strap 60–110 cm; reversible left/right |
| Weight | ≈ 260–320 g depending on size |
| Breathability (air perm.) | ≥ 300 mm/s (ASTM D737) |
| Webbing tensile | ≥ 1,000 N (ASTM D5035) |
| Service life | ≈ 12–24 months routine clinical use; hook-and-loop cycle life ≥ 5,000 openings |
| Sizes | S / M / L (hospital kits often stock M/L) |
| Biocompatibility | ISO 10993 skin-contact materials; latex-free |

Process flow and testing
Materials are cut via CNC for consistent strap geometry; seams are bartacked at stress points; aluminum stays are deburred and anodized. Each batch gets: tensile tests on webbing (ASTM D5035), foam compression set (ASTM D3574), air permeability (ASTM D737), and biocompat checks aligned with ISO 10993 guidance. QMS runs under ISO 13485; risk management per ISO 14971. Typical hospital cycle: pre-fit in PACU, 2-week follow-up for strap tune, and gradual wean in week 4–6—injury depending.

Vendor comparison (quick snapshot)
| Vendor | Certs | Lead Time | Customization | Unit Price (FOB, ≈) |
|---|---|---|---|---|
| JH Orthopedic (Eblow Brace) | ISO 13485; ISO 10993 materials; CE marking support | 15–25 days | Branding, sizes, strap geometry, OEM | $12–$22 |
| Generic A | ISO 9001 only | 25–35 days | Limited colors | $9–$15 |
| Generic B | ISO 13485; no biocomp dossier | 20–30 days | Logo only | $11–$19 |
Use cases: rotator cuff post-op, clavicle strains, humeral fractures (non-displaced), and sideline immobilization. Clinics like the quick-clip strap map; home users like that the sling cradle doesn’t bite into the neck. As a shoulder brace support arm sling it’s meant to immobilize, but it’s forgiving during dressing and shower setup—small detail, big compliance.
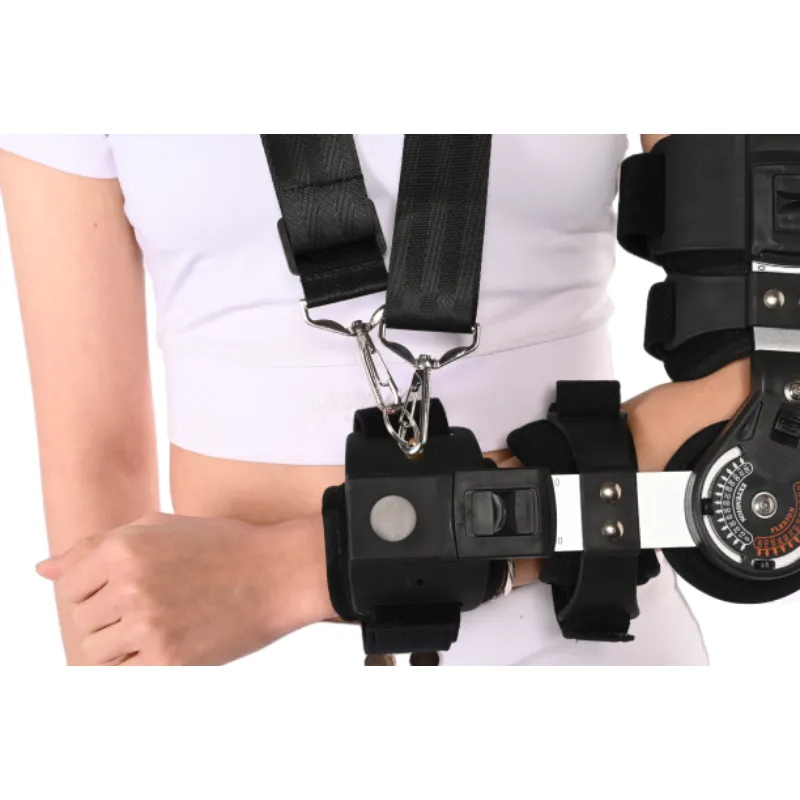
Customization and deployment
OEM options include antimicrobial liners (AATCC 100 target ≥ 99% reduction), colorways for departments, and pediatric sizing. Packaging can be sterile or bulk non-sterile depending on your pathway. For distributors, MOQ is flexible; for hospital systems, forecast-based releases cut stockouts.
Case notes from the field
Sports clinic: A mid-season shoulder subluxation patient used the brace in a strict shoulder brace support arm sling mode for 10 days; reported 30–40% less neck fatigue versus their previous sling (subjective, n=1, but echoed by staff).
Community hospital: Post-op rotator cuff cohort (n=18) noted fewer strap readjustments during rounds; nurses cited clearer strap routing, which—honestly—saves minutes that add up.

Certifications and compliance
Manufactured under ISO 13485 QMS; materials vetted to ISO 10993 for skin contact; risk files align with ISO 14971. Device classification and claims should follow local regulations (e.g., FDA Class I, general controls in the U.S., CE under MDR rules in the EU). Always verify your market’s labeling and UDI needs.
Citations
- ISO 13485: Medical devices — QMS for regulatory purposes. https://www.iso.org/standard/59752.html
- ISO 10993-1: Biological evaluation of medical devices. https://www.iso.org/standard/68936.html
- ASTM D5035: Standard Test Method for Breaking Force and Elongation of Textile Fabrics. https://www.astm.org/d5035
- FDA: Overview of Device Regulation (Class I general controls). https://www.fda.gov/medical-devices/overview-device-regulation
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















