Shoulder Pain Belt Price | Adjustable Compression Support
Shoulder Abduction Orthosis: what affects shoulder pain belt price today
Let me cut to the chase: price isn’t just a number on a product page. It’s the sum of materials, testing, certifications, supply chain sanity, and—frankly—how comfortable the thing feels at 2 a.m. when your shoulder aches. I visited the facility in Anping County, Hebei (No.240 Xingying West Street, for the map lovers) and talked shop with the team behind this Shoulder Abduction Orthosis. Actually, the tech under the hood is more serious than most expect.
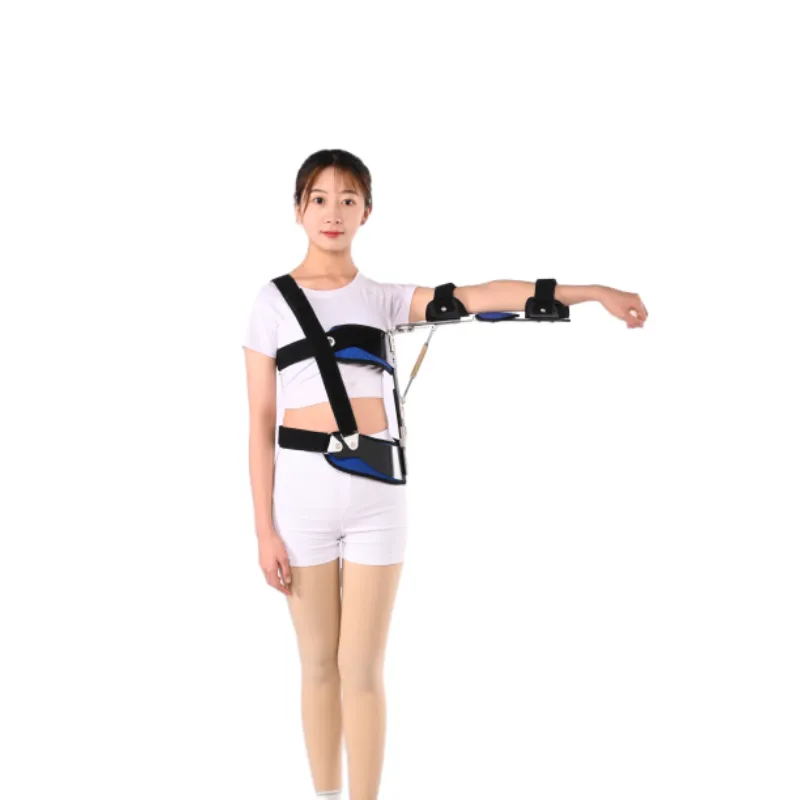
What’s driving the market right now
Three trends keep popping up: breathable spacer fabrics replacing old neoprene, lighter injection-molded frames that still hold abducted angles, and higher compliance with medical standards. Hospitals want predictable outcomes; athletes want mobility without re-injury. This is why the shoulder pain belt price range has crept upward for certified models, while generic imports stay temptingly cheap (with the usual caveats).

Spec sheet (real-world use may vary)
| Product | Shoulder Abduction Orthosis |
| Abduction angles | 30°, 45°, 60°, 90° (adjustable) |
| Materials | Breathable spacer fabric, medical-grade foam, hook-and-loop, injection-molded ABS, aluminum stays |
| Weight | ≈ 420–520 g |
| Testing | ISO 22523 fatigue/structural; ISO 10993 biocompatibility; strap cycle ≥50,000; slippage <5 mm @ 100 N |
| Service life | 12–24 months typical clinic rotation |
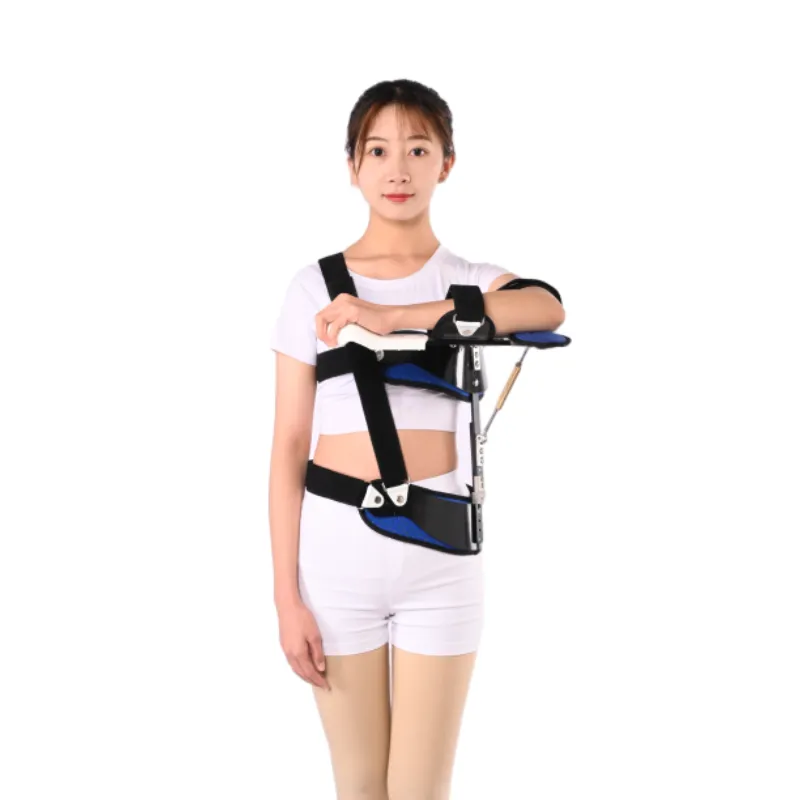
Process flow (why it matters for cost)
Materials are CNC-cut for tight tolerances; pressure zones get bartack stitching; molded ABS shells go through ultrasonic edge finishing. Then there’s QA: tensile tests on straps, cushion compression set checks, and repeated-angle lock testing (yep, it’s as tedious as it sounds, but it weeds out weak units). Certifications—ISO 13485 QMS, CE MDR compliance, FDA class listings—add cost but reduce clinical risk, which ultimately stabilizes the shoulder pain belt price.
Applications, in plain English
Rotator cuff repair (post-op), Bankart lesion rehab, proximal humerus fracture recovery, adhesive capsulitis protocols, and even return-to-work scenarios where abduction control prevents bad habits. Many therapists tell me patients actually keep these on longer because modern pads don’t roast the skin—small thing, big compliance win.
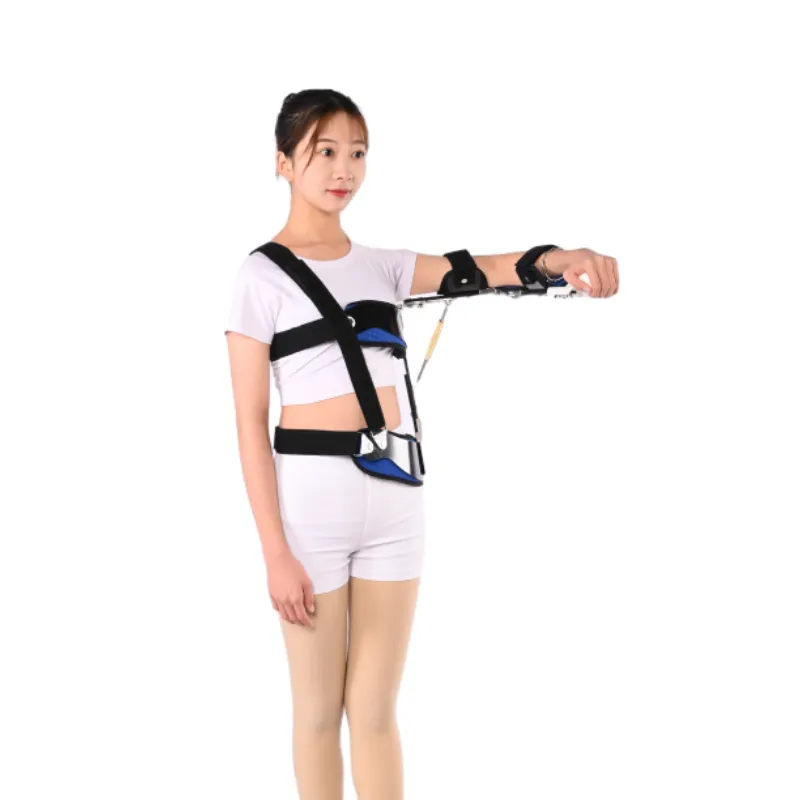
Vendor snapshot (indicative)
| Vendor | Country | Price (USD) | MOQ | Certs | Lead time |
|---|---|---|---|---|---|
| J&H Orthopedic (Anping) | China | ≈ $22–$45 ex-works | 50–100 | ISO 13485, CE MDR | 12–20 days |
| Brand A (US/EU) | US/EU | $80–$160 retail | 1 | FDA, CE | In stock |
| Generic Import | Mixed | $12–$25 | 100+ | Varies | 15–30 days |
Note: ranges are indicative; specs, freight, and compliance can shift the final shoulder pain belt price.
Customization options
OEM colors, private labeling, left/right configurations, pediatric sizing, antimicrobial textiles, and angle locks set at 30°/45°/60° out of the box. Hospitals often ask for washable liners and barcode tracking—nice touches that don’t break the bank.
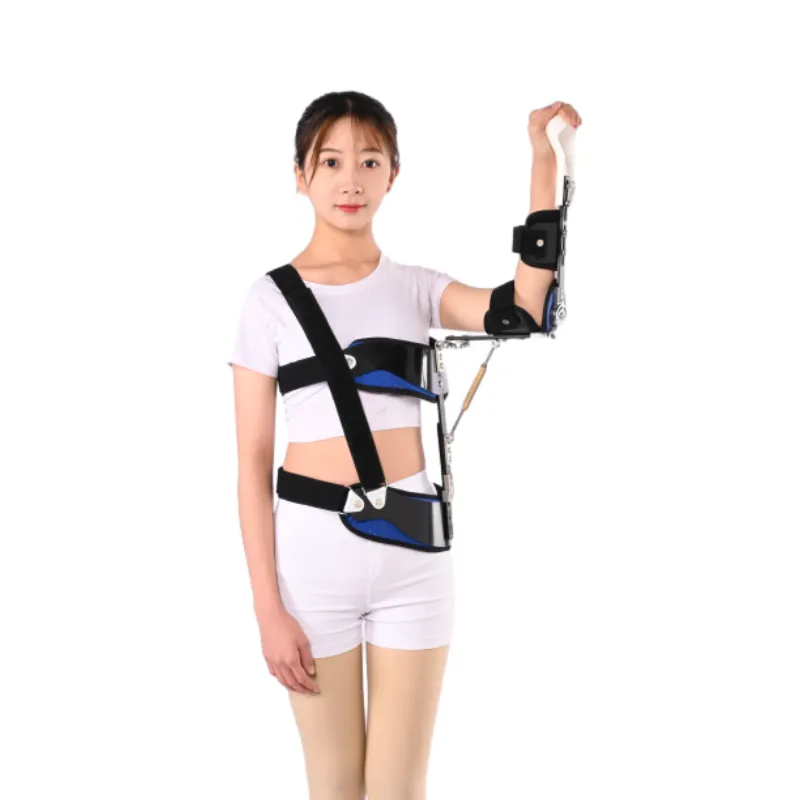
Real users, quick cases
Case 1: Post–rotator cuff repair, 54-year-old teacher. Reported “less neck bite” and slept through the night by week two. Therapist noted improved adherence due to breathable liner.
Case 2: Amateur tennis player, Bankart repair. Kept at 45° abduction for four weeks; sling stability under commuting jostle was “surprisingly solid,” according to him.
Bottom line
If you’re benchmarking, focus on certified materials, adjustable abduction, liner breathability, and verified test data. When those boxes are ticked, the shoulder pain belt price tends to make sense—because cheaper failures cost more in the clinic, and in patience.
Authoritative sources
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















