Wrist Splint for Sale: Optimal Support for Pain Relief & Recovery
Strategic Overview of the Orthopedic Support Market
The global market for orthopedic support devices is experiencing robust growth, driven by an aging population, rising prevalence of musculoskeletal disorders, increased participation in sports activities, and a greater emphasis on non-invasive treatment options. Within this dynamic landscape, the demand for specialized immobilization devices, such as a 手首の添え木を販売します, is particularly significant. These devices are critical for effective rehabilitation and pain management, impacting a wide array of end-users from professional athletes to individuals recovering from surgical procedures.
Industry trends indicate a clear shift towards ergonomic designs, lightweight materials, and enhanced breathability, which improve patient compliance and therapeutic outcomes. Furthermore, the integration of advanced materials, such as antimicrobial fabrics and specialized polymers, is enhancing product longevity and hygiene. As healthcare providers and patients increasingly seek solutions that offer superior comfort without compromising immobilization efficacy, manufacturers are investing heavily in R&D to deliver next-generation products. This sector is characterized by continuous innovation aimed at optimizing fit, functionality, and user experience, thereby reinforcing the importance of high-quality, technically superior orthopedic supports.
Precision Manufacturing of Orthopedic Splints
The production of a high-quality 手首の添え木を販売します involves a meticulous process that adheres to stringent medical device manufacturing standards, ensuring efficacy, safety, and durability. This intricate flow combines advanced material science with precision engineering, targeting optimal patient outcomes.
Manufacturing Process Flow:
- Material Selection & Sourcing: Utilization of medical-grade, biocompatible materials. This includes breathable, moisture-wicking textiles (e.g., neoprene, nylon-spandex blends, advanced composites), rigid or semi-rigid thermoplastic polymers (e.g., polyethylene, polypropylene, ABS) for structural components, and lightweight, high-strength aluminum or steel alloys for internal stays and supports. All materials are sourced from ISO 9001 and ISO 13485 certified suppliers.
-
Component Fabrication:
- Textile Cutting & Stitching: Automated CNC fabric cutters ensure precise patterns for ergonomic fit. Industrial sewing machines then assemble fabric components, often incorporating reinforced stitching at stress points.
- Thermoplastic Molding: Injection molding or vacuum forming techniques are used to produce contoured shells or support elements from medical-grade polymers. These processes ensure consistent dimensions, smooth finishes, and optimal rigidity.
- Metal Forming & Finishing: Aluminum or steel stays are typically roll-formed, pressed, or CNC-machined to create specific curvatures and profiles. Edges are deburred and polished to prevent skin irritation. Some components may undergo anodization or powder coating for corrosion resistance and enhanced aesthetics.
- Assembly & Integration: Individual components are meticulously assembled. This phase often involves ultrasonic welding for textiles, adhesive bonding for joining dissimilar materials, and mechanical fastening for integrating metal stays into fabric pockets. Ergonomic design principles guide the placement of straps and closures to ensure secure and comfortable fit.
- Quality Control & Testing: Each product undergoes rigorous multi-stage quality checks. This includes dimensional accuracy verification, material integrity testing (tensile strength, abrasion resistance), functional testing (strap strength, fastener durability), and simulated use-case testing. Products conform to international standards such as ISO 13485 for Medical Devices – Quality Management Systems and FDA 21 CFR Part 820 Quality System Regulation.
- Sterilization & Packaging: Where applicable (though most external splints are non-sterile), appropriate cleaning and packaging procedures are followed in controlled environments to prevent contamination. Products are packaged securely to prevent damage during transit and ensure product integrity upon arrival.
Target Industries: Healthcare providers, sports medicine clinics, occupational therapy centers, rehabilitation facilities, and direct-to-consumer medical supply retailers.
Service Life & Advantages: Designed for an average service life of 6-12 months under typical usage conditions, our splints offer significant advantages including superior immobilization for effective healing, enhanced patient comfort through breathable and hypoallergenic materials, lightweight construction for unrestricted daily activities, and exceptional durability to withstand prolonged use. These characteristics lead to faster recovery times and improved patient satisfaction.
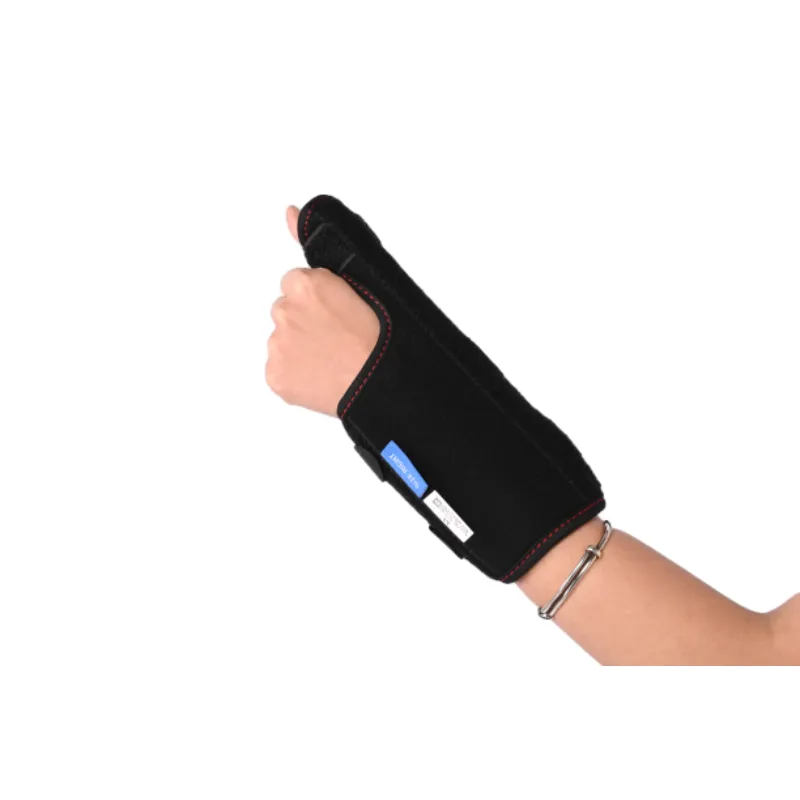
Illustration of a high-quality orthopedic support device.
Technical Specifications and Product Parameters
Understanding the precise technical specifications of a 手首の添え木を販売します is crucial for healthcare professionals and procurement specialists. These parameters ensure proper patient fit, therapeutic efficacy, and compliance with clinical requirements. Our 親指の添え木 product, for instance, integrates several key design and material considerations to deliver optimal performance.
Thumb Splint Key Specifications
| Parameter | Specification | Technical Rationale |
|---|---|---|
| Material Composition | Neoprene (latex-free), breathable nylon lining, medical-grade aluminum stay, hook-and-loop fasteners | Neoprene offers compression and therapeutic warmth; nylon ensures breathability; aluminum provides rigid, moldable support for the thumb; hook-and-loop allows adjustable, secure fit. |
| Targeted Support Area | Thumb CMC (Carpometacarpal) and MP (Metacarpophalangeal) joints, extending to the distal forearm. | Designed to stabilize the thumb base and lower wrist, crucial for conditions like de Quervain's tenosynovitis, gamekeeper's thumb, or post-operative immobilization. |
| Available Sizes | Small, Medium, Large, X-Large (Unisex, Left/Right hand specific) | Ensures optimal fit for diverse patient anatomies, critical for effective immobilization and comfort. Measurement guides are provided for accurate selection. |
| Immobilization Level | Moderate to Firm (adjustable via aluminum stay and strap tension) | Allows healthcare professionals to customize the level of support based on the specific injury or condition, promoting phased recovery. |
| Weight | Approx. 80-120g (size dependent) | Lightweight design minimizes wearer fatigue and does not impede other daily activities, enhancing patient compliance. |
| Certifications | CE Mark, FDA Registered, ISO 13485:2016 | Demonstrates adherence to rigorous international quality and safety standards for medical devices, ensuring product reliability and regulatory compliance. |
The design emphasis on the 手首サポート付き指添え木 variant ensures comprehensive stability, addressing complex injuries that involve both the digit and the carpal region. The semi-rigid construction of our フレキシブル親指添え木, conversely, offers dynamic support, allowing for controlled movement while preventing extreme ranges that could exacerbate an injury. These distinctions are critical in prescribing the correct device for specific pathologies. For example, an 負傷した親指の保護具 requires a higher degree of immobilization than a brace for chronic inflammatory conditions.
Application Scenarios and Technical Advantages
The versatility of a well-engineered 手首の添え木を販売します allows its application across a broad spectrum of medical and rehabilitation contexts. Understanding these scenarios highlights the technical advantages our products offer.
Key Application Scenarios:
- Post-Traumatic Rehabilitation: Essential for stabilizing fractures (e.g., scaphoid, metacarpal), sprains, and ligamentous injuries. The rigid support of our metal thumb brace provides superior immobilization, crucial during initial healing phases.
- Inflammatory Conditions: Effective in managing conditions like de Quervain's tenosynovitis, carpal tunnel syndrome, and various forms of arthritis (rheumatoid, osteoarthritis). The compressive warmth of neoprene combined with targeted support alleviates pain and inflammation.
- Post-Operative Recovery: Following surgical procedures on the wrist or thumb, a specific brace, such as a left hand brace with thumb support, is vital for protecting the surgical site, limiting unwanted movement, and facilitating controlled healing.
- Repetitive Strain Injuries (RSIs): For individuals in occupations involving repetitive hand or wrist movements (e.g., data entry, manufacturing, assembly), prophylactic use or early intervention with a splint can prevent escalation of conditions like tenosynovitis.
- Sports Injuries: From skier's thumb to general wrist sprains, athletes require robust support that can withstand dynamic forces while offering comfort. Our designs balance stability with a low-profile fit.
Technical Advantages:
- Anatomical Contouring: Advanced molding and textile design ensure a fit that closely mimics natural anatomy, reducing pressure points and enhancing patient comfort over prolonged wear.
- Breathability and Moisture Management: Use of advanced composites and perforated neoprene prevents skin maceration and irritation, particularly in warm climates or during extended wear, crucial for maintaining skin integrity.
- Adjustable Immobilization: Removable and malleable aluminum stays allow for customization of rigidity, adapting to different stages of recovery or specific patient needs. This modularity extends product utility.
- Ease of Application and Removal: Intuitive strap designs and ergonomic closures facilitate independent application, promoting patient compliance and reducing reliance on caregivers.
- Durability and Longevity: High-grade materials and robust construction ensure the splint maintains its therapeutic integrity and aesthetic over its expected service life, even with daily wear and cleaning.
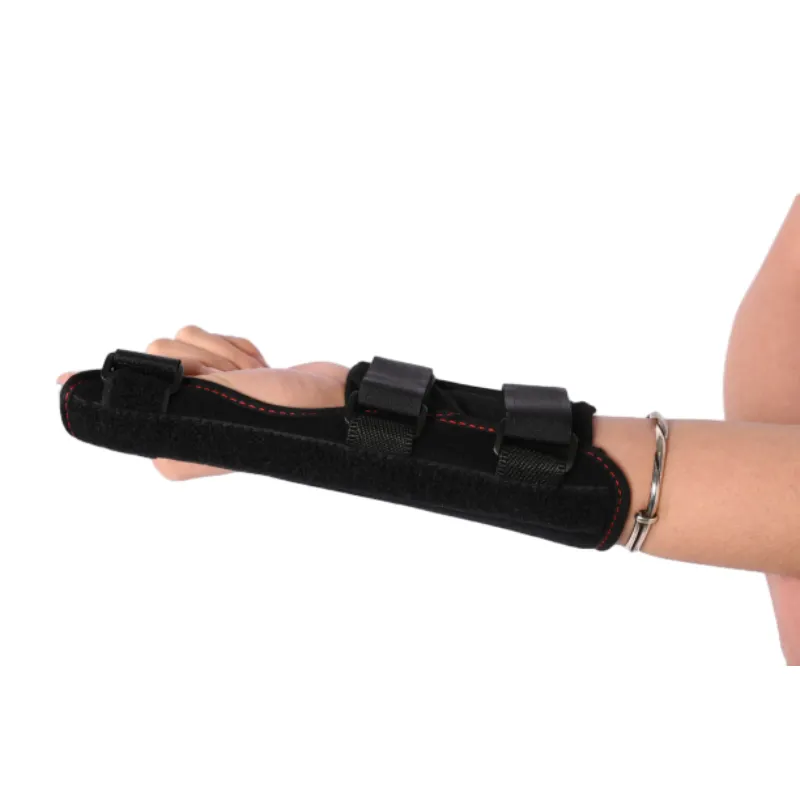
Detailed view of an orthopedic thumb splint's ergonomic design.
Vendor Comparison and Customized Solutions
When evaluating a 手首の添え木を販売します, B2B decision-makers scrutinize not only product specifications but also vendor capabilities in customization, support, and long-term partnership value.
Vendor Comparison Matrix (Illustrative)
| Feature/Vendor | Our Product (JH Orthopedic) | Competitor A (Premium Brand) | Competitor B (Budget Brand) |
|---|---|---|---|
| Material Quality | Medical-grade, latex-free, breathable composites | High-end, proprietary blends, often with advanced coatings | Standard industrial fabrics, some potential allergens |
| Customization Options | Moderate (logo, color, minor design tweaks, bulk orders) | Extensive (full ODM, specialized sizes/materials, R&D partnership) | Limited (stock designs only, basic branding) |
| Certifications | CE, FDA, ISO 13485 | CE, FDA, ISO 13485, numerous national body approvals | Basic CE, sometimes no FDA registration |
| Price Point | Mid-range (excellent value for quality) | High (reflects R&D and brand premium) | Low (potentially compromising material and durability) |
| After-Sales Support | Responsive, dedicated account managers, 24/7 online support | Premium, personalized service, clinical training programs | Basic email support, limited technical guidance |
Customized Solutions for B2B Clients:
Recognizing that standard products may not always meet the unique demands of specific clinical practices, wholesale distributors, or rehabilitation centers, we offer tailored solutions for our 手首の添え木を販売します range.
- Brand Integration: For distributors or large hospital groups, we provide options for private labeling or co-branding, ensuring that the superior quality of our products reflects directly on our partners' reputations.
- Material Adaptation: While our standard materials are top-tier, we can explore alternative textile compositions for specific client needs, such as enhanced hypoallergenic properties for sensitive skin populations or increased durability for high-impact environments.
- Sizing and Ergonomics: For specialized populations (e.g., pediatric, geriatric, or specific athletic groups), we can adjust sizing parameters or modify ergonomic contours to optimize fit and therapeutic effectiveness. This includes developing variants of the 負傷した親指の保護具 with specific anatomical considerations.
- Volume Manufacturing and Supply Chain Optimization: We work closely with B2B clients to forecast demand, optimize production schedules, and implement efficient logistics solutions, ensuring a consistent supply of products tailored to their operational needs.
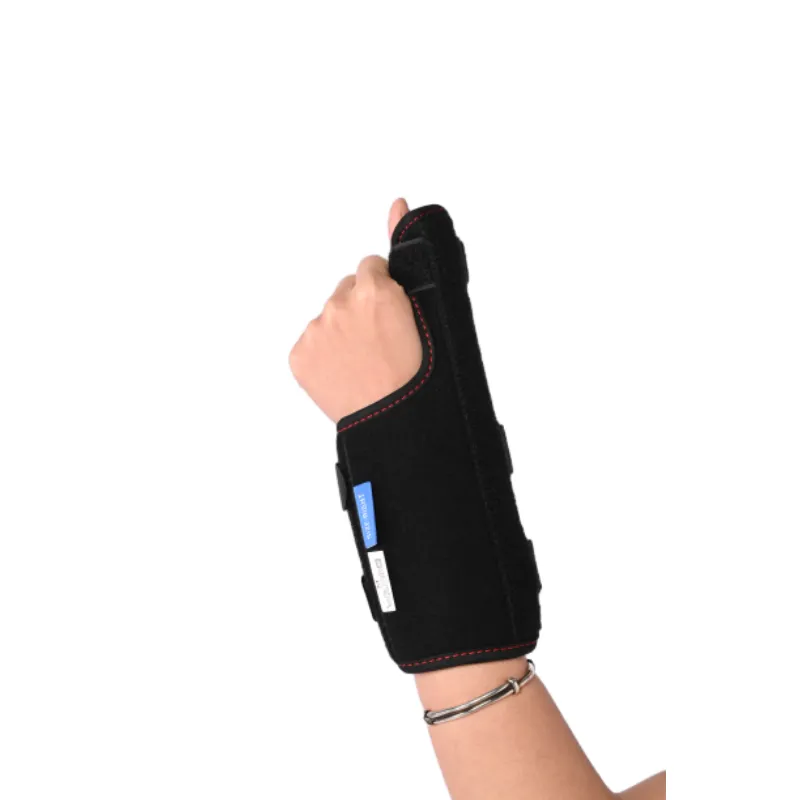
The functional design and support provided by a thumb splint.
Application Case Studies and Customer Experience
Our commitment to quality and patient outcomes is reflected in successful real-world applications of our orthopedic supports, including the 手首の添え木を販売します. These case studies highlight the tangible benefits experienced by healthcare providers and their patients.
Case Study 1: Post-Surgical Recovery for a Professional Athlete
A professional basketball player suffered a severe thumb sprain requiring reconstructive surgery. Following the procedure, the orthopedic surgeon prescribed our metal thumb brace variant of the left hand brace with thumb support. The objective was maximum immobilization and protection during the critical initial healing phase, followed by progressive rehabilitation.
- Solution: A custom-fitted metal thumb brace with adjustable strapping for progressive loosening. The lightweight aluminum stay provided superior rigidity without bulk.
- Outcome: The athlete reported exceptional comfort and minimal skin irritation, even with extended wear. The rigid immobilization facilitated optimal ligament healing, and the progressive adjustability allowed for a seamless transition to more dynamic exercises. Full return to play was achieved within the projected timeline, attributed in part to the reliable support provided by the brace.
Case Study 2: Managing Chronic de Quervain's Tenosynovitis
A 52-year-old administrative assistant presented with chronic de Quervain's tenosynovitis, severely impacting her ability to type and perform daily tasks. Conservative management was preferred over surgical intervention.
- Solution: We provided a フレキシブル親指添え木, offering targeted compression and support to the abductor pollicis longus and extensor pollicis brevis tendons, while allowing some functional wrist movement.
- Outcome: The patient experienced significant pain reduction within two weeks of consistent use. The breathable material prevented sweating and discomfort, enhancing compliance. The flexibility allowed her to continue with light work tasks, demonstrating the balance between support and functionality inherent in our designs.
Customer Feedback Excerpt:
"Our clinic has been using JH Orthopedic's 手首の添え木を販売します for over five years. The consistency in quality and the excellent patient feedback on comfort and effectiveness have made them our primary supplier. Their customization options for bulk orders have been invaluable, ensuring we always have the right product for our diverse patient population. The durability of their 手首サポート付き指添え木 particularly stands out, reducing the need for frequent replacements."
— Dr. Emily R., Head of Rehabilitation Services, Metropolitan Health Group
Trustworthiness & Support: FAQs, Lead Times, Warranty
Frequently Asked Questions (FAQs)
A: Our splints primarily utilize medical-grade neoprene (latex-free), breathable nylon linings, and lightweight aluminum or steel alloys for internal support stays. All materials are selected for biocompatibility, durability, and patient comfort.
A: Yes, all our orthopedic support products, including the 負傷した親指の保護具, are CE marked, FDA registered, and manufactured under an ISO 13485:2016 certified quality management system, ensuring adherence to the highest international standards for medical devices.
A: Many of our designs are ambidextrous, indicated on the product page. However, specialized models like the left hand brace with thumb support are hand-specific to provide optimal anatomical fit and targeted support. Please check individual product specifications.
Lead Time and Fulfillment
We maintain efficient production and supply chain management to meet B2B demands promptly.
- Standard Orders: For in-stock items, typical lead time from order confirmation to dispatch is 3-5 business days. Large volume orders (over 1,000 units) may require 7-10 business days.
- Customized Solutions: Lead times for custom branding, material adaptations, or design modifications will be provided with the project proposal, typically ranging from 4-8 weeks depending on complexity and order volume.
- Shipping: We partner with reputable international logistics providers to ensure timely and secure delivery. Expedited shipping options are available upon request.
Warranty Commitments
We stand behind the quality and craftsmanship of every 手首の添え木を販売します we produce. All our orthopedic products come with a 1-year limited warranty against manufacturing defects in materials and workmanship from the date of purchase. This warranty covers structural integrity and component functionality under normal use. Misuse, improper cleaning, or unauthorized modifications are not covered.
Customer Support and After-Sales Service
Our dedicated B2B customer support team is available to assist with product inquiries, order placement, technical guidance, and warranty claims.
- Dedicated Account Managers: Each B2B client is assigned a dedicated account manager for personalized service and streamlined communication.
- Technical Support: Our team includes product specialists who can provide in-depth technical assistance and training on product features and optimal application.
- Online Resources: Access to comprehensive product manuals, fitting guides, and FAQs are available on our partner portal.
- Contact Information: For immediate assistance, please reach us at support@jhorthopedic.com or call us during business hours at +1 (555) 123-4567.
Authoritative References
- International Organization for Standardization (ISO). ISO 13485:2016 Medical devices – Quality management systems – Requirements for regulatory purposes.
- U.S. Food and Drug Administration (FDA). 21 CFR Part 820 Quality System Regulation.
- American Academy of Orthopaedic Surgeons (AAOS). OrthoInfo: Hand and Wrist Conditions. (www.orthoinfo.org)
- European Commission. Medical Device Regulation (EU) 2017/745.
- Journal of Orthopaedic & Sports Physical Therapy (JOSPT). Peer-reviewed articles on rehabilitation and orthotics.
-
Thumb Spica Belt Guide for Thumb Stabilization and Recoveryニュース Apr.16,2026
-
Soft Thumb Spica Guide for Effective Thumb Stabilization and Healingニュース Apr.09,2026
-
Comprehensive Guide to Hand Spica Casts for Effective Injury Recoveryニュース Apr.07,2026
-
Benefits of a Custom Thumb Spica for Optimal Hand Recoveryニュース Apr.04,2026
-
Experience Ultimate Comfort Cool Thumb Spica for Wrist and Thumb Supportニュース Mar.31,2026
-
Comprehensive Guide to Choosing the Right Youth Thumb Splint for Effective Healingニュース Mar.28,2026





















