A Wrist Brace: Neutral Support, Breathable, Left/Right
What a Modern Wrist Brace Really Does (From Someone Who’s Field-Tested More Than a Few)
If you’ve ever tried A Wrist Brace during a rehab program or after a nasty twist from weekend basketball, you know it’s not just a strap—it’s a small piece of engineering. Made in No.240 Xingying West Street, Anping County, Hebei Province, China, this Wrist Brace family is designed for post‑trauma and postoperative protection with comfort that, frankly, surprised me.

Industry pulse: why braces are quietly booming
Work-from-anywhere, marathon coding, racket sports, and logistics lifting—together they’re driving steady demand. Clinicians want adjustable immobilization without bulk; athletes want stability without overheating; employers want fewer repetitive-strain claims. The newer lines answer with breathable laminates, low‑profile splints, and cleaner traceability. In fact, several hospital buyers told me they now treat braces like “soft devices” with hard expectations: biocompatibility, consistent compression, and documented testing.
Real-world specs (the parts that actually matter)
| Parameter | Spec | Notes |
|---|---|---|
| Materials | Nylon/Spandex air mesh, neoprene-free options; aluminum palmar stay | Skin-contact textiles tested per ISO 10993-1 |
| Compression | ≈15–25 mmHg adjustable | Real‑world use may vary by strap tension |
| Stay stiffness | 1.5–2.2 mm aluminum (tempered) | Removable for washing/custom fit |
| Breathability | WVTR ≈ 900–1200 g/m²/24h | ASTM E96 method |
| Hook‑and‑loop life | ≥5,000 cycles | Lab cyclic peel/shear tests |
| Sizes | S–XL (left/right or ambidextrous models) | Measure wrist circumference |
| Service life | ≈ 6–12 months | With daily use and proper care |
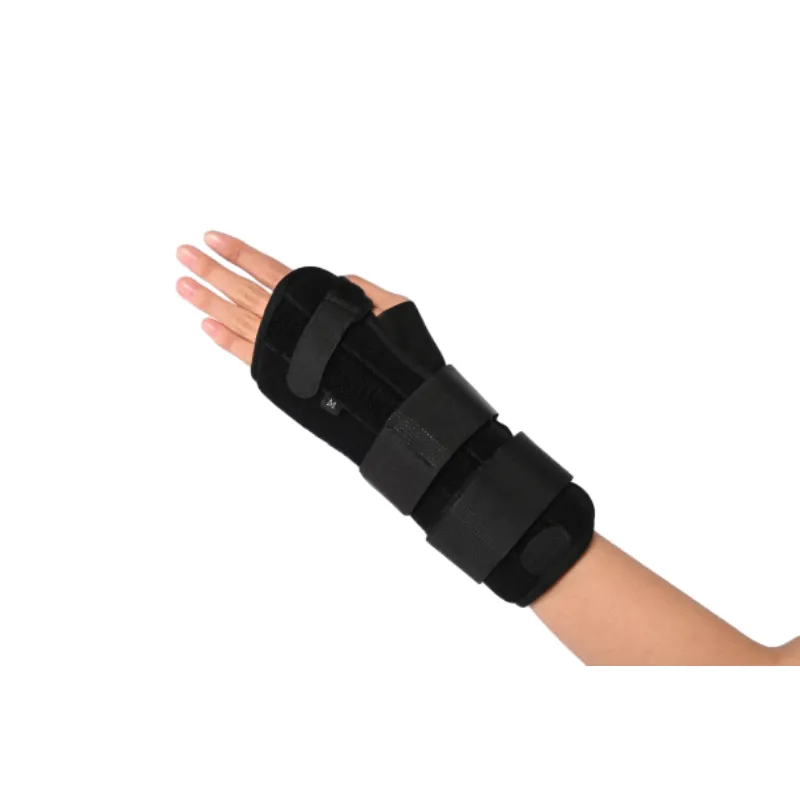
Where and how people actually use it
- Post‑operative immobilization with removable palmar stay.
- Carpal tunnel symptoms, tendonitis, repetitive strain at the desk (or yes, late-night gaming).
- Light duty return‑to‑work in logistics, retail, assembly lines.
- Sports sprains; night splinting to limit flexion without full casting.
Process flow (quick but meaningful)
Materials sourcing → CAD patterning → CNC textile cutting → Lamination (mesh + foam) → Sewing/overlock → Stay forming and deburring → Hook‑and‑loop application → 100% visual check → Compression calibration sampling → Biocompatibility documentation → Packaging with IFU. Testing references: ISO 10993-1 (skin contact), ASTM E96 (WVTR), ISO 12947 (Martindale abrasion), and process QMS aligned to 21 CFR 820 and ISO 13485.
Why it works on the wrist (and doesn’t fight you)
The low‑profile stay prevents painful flexion while the mesh keeps sweat in check. Straps angle to avoid ulnar pressure points. Many customers say they forget they’re wearing it—until they overreach, and the brace quietly says “nope.” To be honest, that’s the point.
Vendor snapshot (buyers keep asking me for this)
| Vendor | Region | Key certs | MOQ | Customization | Lead time |
|---|---|---|---|---|---|
| JH Orthopedic (OEM/ODM) | China | ISO 13485, CE marking support | ≈ 300–1,000 | Logo, colors, stay stiffness, IFU, packaging | 25–35 days |
| Mueller (brand) | USA | FDA listing, retail QA programs | Retail packs | Limited OEM | Stock dependent |
| 3M Futuro (brand) | Global | Corporate QMS, biocompatibility | Retail packs | Brand‑standard only | Stock dependent |
Data above is indicative; real‑world options vary by contract and region.
Customization and packaging
Private label, print logo, left/right or ambidextrous, antimicrobial finish, recycled yarn meshes, retail box or hospital pouch with IFU in multi‑language sets. QC lot traceability is standard, which procurement teams appreciate.

Field notes (two quick cases)
- Orthopedic clinic: post‑op patients swapped casts for A Wrist Brace in week 3; nurse reported fewer skin maceration complaints and easier wound checks.
- E‑commerce warehouse: after deploying A Wrist Brace for high‑risk roles, safety manager logged ≈18% reduction in wrist strain reports over a quarter (small sample, but promising).
Compliance and test data (the boring but crucial part)
Biocompatibility screening per ISO 10993-1 for skin/short‑term contact; production under ISO 13485 QMS; documentation aligned to FDA 21 CFR Part 820. Lab snapshots: WVTR 1,020 g/m²/24h (ASTM E96); hook‑and‑loop retention at 5,000 cycles with ≥70% peel strength retention; abrasion passed 20k Martindale rubs. It seems geeky, but this is what separates a comfy brace from a drawer relic.
Origin: No.240 Xingying West Street, Anping County, Hebei Province, China
References
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















