Thoracic Lumbar Back Support - Adjustable, Lightweight
Field Notes on a Modern Thoracic Lumbar Back Support
I’ve been around orthopedics long enough to see spinal braces evolve from bulky, sweat-inducing shells to streamlined devices you can (almost) forget you’re wearing. The Thoracolumar Brace—built in Anping County, Hebei—lands right in that sweet spot: rigid where it matters, forgiving where it should be. In fact, the broader market is shifting toward lighter composites, breathable liners, and smarter strap systems. To be honest, that’s overdue.

What it is and where it fits
Officially, this is a Thoracolumbar Sacral Orthosis (TLSO). Clinically, it’s used post-op, after thoracic/lumbar fractures, during conservative care for degenerative changes, and sometimes for posture retraining. Many customers say the dual-pull tightening is the game changer—quick to don, easy to adjust mid-day. I guess that sounds minor until your back twinges at 3 p.m.

Technical specifications (real-world use may vary)
| Model | Thoracolumar Brace (TLSO) |
| Coverage | Approx. T6–L5 stabilization, adjustable |
| Materials | ABS/PP shells, 6061 Al uprights, EVA foam pads, breathable spacer fabric |
| Closure | Dual-pull mechanical-advantage straps, hook-and-loop |
| Weight | ≈ 0.9–1.2 kg depending on size |
| Sizes | S–XXL, trim-to-fit panels |
| Service life | ≈ 12–24 months under typical clinical use |
| Standards (targeted) | ISO 22523, ISO 10993 for skin-contact materials, QMS per ISO 13485 |
Origin: No.240 Xingying West Street, Anping County, Hebei Province, China.

How it’s made (short version)
- Materials: ABS/PP shells vacuum-formed; aluminum uprights CNC-trimmed; liners in moisture-wicking spacer fabric.
- Methods: Heat-forming, precision trimming, riveted joints, bar-tack stitching on high-load points.
- Testing: Strap cycle endurance ≥ 10,000 pulls (internal bench test, n=3); shell flexural checks; biocompatibility per ISO 10993 guidance; orthosis requirements aligned to ISO 22523.
- QA: Incoming material inspection, torque verification on uprights, fit range checks, documentation under ISO 13485 procedures.

Where it’s used (and by whom)
Post-op fusion (T10–L4), osteoporotic compression fractures, degenerative disc disease flare-ups, and—surprisingly—some light industrial settings for short-term lift support under clinician advice. One hospital buyer told me their staff like the fast adjustability during discharge fitting. Another user said the liner “doesn’t feel like a sauna,” which is nice.
Vendor landscape (quick compare)
| Vendor | Origin | Lead time | Customization | Certs (typical) | Price index |
|---|---|---|---|---|---|
| JH Orthopedic (Thoracolumar Brace) | China | ≈ 2–4 weeks | Shell color, logo, sternum pad, sizing tweaks | ISO 13485, ISO 10993 (verify per lot) | $ (value) |
| US-based OEM | USA | ≈ 3–6 weeks | Broader component library | ISO 13485, FDA listing (check) | $$$ |
| EU distributor | EU | Stock/2–3 weeks | Limited (branding, sizes) | CE marking (verify) | $$ |

Customization and options
Typical mods include removable sternum pad for kyphosis control, extended lumbar plate, hypoallergenic liner, and company logo/colorways. For clinics, batch labeling and barcodes help with inventory. Small thing, big difference.
Mini case files
- Post-op fusion patient: earlier upright tolerance by day 3; nurse noted faster donning versus legacy brace.
- Warehouse supervisor: intermittent use during return-to-work; reported less mid-day fatigue with Thoracic Lumbar Back Support than soft corsets.
- Osteoporotic VCF: short-term stabilization; PT liked the adjust-on-the-fly straps for graded activity.
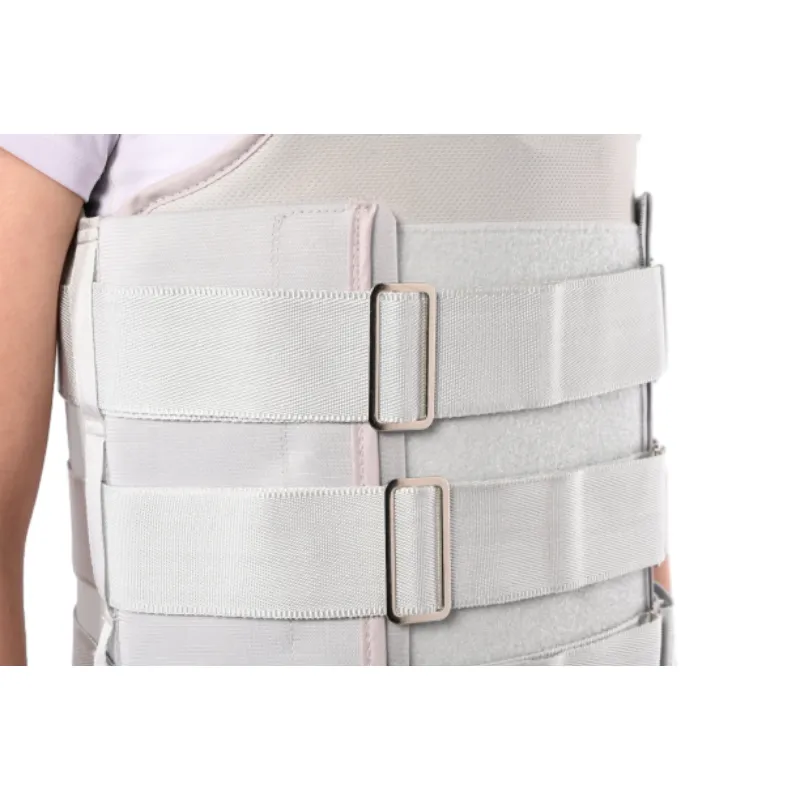
Why it matters
Rigid bracing isn’t glamorous. But the right Thoracic Lumbar Back Support can stabilize healing segments, reduce painful micromotion, and make daily life tolerable while the spine recovers. Always fit and prescribe under clinician guidance—yes, I’m repeating myself on purpose.
References
- ISO 22523:2022 External limb prostheses and orthoses — Requirements and test methods.
- ISO 10993-1:2018 Biological evaluation of medical devices — Part 1.
- ISO 13485:2016 Medical devices — Quality management systems — Requirements.
- North American Spine Society. Clinical Guidelines for Thoracolumbar Spine Trauma and Bracing.
- ASTM D638. Standard Test Method for Tensile Properties of Plastics.
-
Comprehensive Guide to Making Arm Sling – Durable Orthopedic Solutions for B2B BuyersNews Jan.05,2026
-
What Is A Arm Sling Used For – Key Insights from JH OrthopedicNews Dec.11,2025
-
What Injuries Need An Arm Sling – Industry Overview & Best Choices for B2B BuyersNews Dec.11,2025
-
What Are Arm Slings Used For? Benefits and Vendor ComparisonNews Dec.11,2025
-
Purpose of Arm Sling | Support & Recovery EssentialsNews Dec.10,2025
-
Making A Sling For Your Arm: Durable, Comfortable Solutions for Medical ProfessionalsNews Dec.10,2025





















